Controversies in Perianal Fistulizing Crohn’s Disease: Debunking the Myths
Controversies in Perianal Fistulizing Crohn’s Disease: Debunking the Myths
April 28, 2026
Issue 04
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by Dr. Jeffrey McCurdy, at the 26th annual national meeting, Mentoring in IBD XXVI: The Master Class, held in Toronto on November 14, 2025.
Introduction
The objectives of this presentation were to present five controversial areas pertaining to the diagnosis, evaluation, and management of perianal fistulizing Crohn’s disease (PFCD), and the latest data to support best practices. The controversial areas include how to screen for PFCD, how to determine the cause of isolated perianal fistulas, the role of combination medical and surgical therapy, efficacy of non-anti-tumor necrosis factor (TNF) therapies, and assessing healing.
Controversy #1: Is A Clinical Exam Sufficient to Rule Out PFCD in Asymptomatic Patients?
Two recent studies challenge the notion that standard physical exams of the perineum are sufficient to rule out PFCD by demonstrating that pelvic Magnetic Resonance Imaging (MRI) can detect subclinical fistulas.
The first study used Magnetic Resonance Enterography (MRE) supplemented with pelvic MRIs in adults diagnosed with or suspected of having CD.1 The second was a Canadian pediatric study evaluating consecutive children with newly diagnosed CD who underwent pelvic MRIs.2
In both studies, subclinical fistulas were detected by MRI imaging. Up to 20% of patients were found to have subclinical fistulas who previously had no history of perianal fistulas, perianal symptoms or visible perianal disease on clinical exam. Those patients with subclinical fistulas were more likely to develop symptomatic disease (adjusted hazard ratio [HR]=3.45) or require perianal surgery (HR=1.40).1,2
Controversy #2: Does Perianal CD Require the Presence of Luminal Inflammation?
In clinical practice, patients may present with perianal fistulas despite a normal ileocolonoscopy. The most common causes for this presentation include cryptoglandular disease and isolated PFCD. Determining the correct diagnosis is critical because treatment differs substantially: surgery alone versus medical therapy with or without surgery.
One potential method to distinguish PFCD from cryptoglandular fistulas is to assess for occult luminal inflammation. In a study published by McCurdy and colleagues,3 45 consecutive patients with perianal fistulas with normal ileocolonoscopy and cross-sectional abdominal imaging underwent video capsule endoscopy (VCE). Inflammation, suggestive of CD, was detected in the small bowel in 26% of patients.3 These findings suggest that VCE may be helpful in differentiating cryptoglandular fistulas from isolated PFCD. However, it should be recognized that perianal fistulas may still be caused by CD even in the absence of luminal inflammation, referred to as “isolated PFCD”.
The TOpClass consortium recently published expert consensus guidelines to provide clinicians with a framework for when to suspect isolated PFCD. These guidelines suggest that isolated PFCD should be suspected when one or more independent criteria (epithelioid granulomas detected by fistula tract biopsies or macroscopic disease) are present or by a composite score of 5 or more points derived from major and minor criteria (Figure 1).4

When the TOpClass criteria is satisfied a trial of anti-TNF therapy is warranted.
Controversy #3: Are Exams Under Anesthesia and Setons Required for All Patients with PFCD?
Traditionally, complex PFCD is treated with a combination of surgical and medical therapies consisting of exam under anesthesia (EUA) with seton(s) and immune-targeted therapies. However, the universal use of EUA and setons is largely based on expert opinion rather than high-quality evidence.
A recent meta-analysis by Fung and colleagues,5 examined whether combined medical and surgical (EUA with or without setons) therapy improves fistula remission. Combined modality therapy was found to be superior to surgery alone (relative risk=1.17; 95% CI, 1.00-1.36; p=0.05), but not to anti-TNF therapy alone (relative risk=1.06; 95% CI, 0.86-1.31; p=0.58). This study had several limitations including a small number of studies with limited sample sizes, lack of treatment randomization, and lack of adjustments for confounding variables.
To further investigate, McCurdy and colleagues6 conducted a multicenter observational study comparing fistula outcomes among 221 patients with PFCD based on the presence or absence of setons at the time of anti-TNF therapy initiation. Outcomes were adjusted for fistula anatomy, complexity, and severity based on pre-treatment pelvic MRI imaging. No significant differences in major adverse fistula outcomes (HR=1.23; 95% CI, 0.68-2.21; p=0.488) or fistula remission (odds ratio=0.81; 95% CI, 0.41-1.59; p=0.54) were observed between with and without setons. However, in a subgroup of patients with abscesses, detected by pre-treatment MRI, setons were associated with lower rates of major adverse fistula outcomes (HR=0.49; 95% CI, 0.19-1.25; p=0.126), although not statistically significant.6 Overall, these findings suggest that setons may not be required for all patients, although randomized controlled trials are needed for further confirmation.
Controversy #4: Is Anti-TNF Therapy the Only Effective Medical Therapy?
To date, infliximab (IFX) is the only medical therapy studied in a dedicated, phase 3 trial for patients with PFCD. As a result, IFX remains the first-line therapy for this phenotype. However, emerging evidence suggests that other advanced therapies might also be effective. Post-hoc subgroup analyses demonstrated higher rates of fistula closure when treated with vedolizumab, ustekinumab, and upadacitinib compared with placebo as summarized in Figure 2.7-9
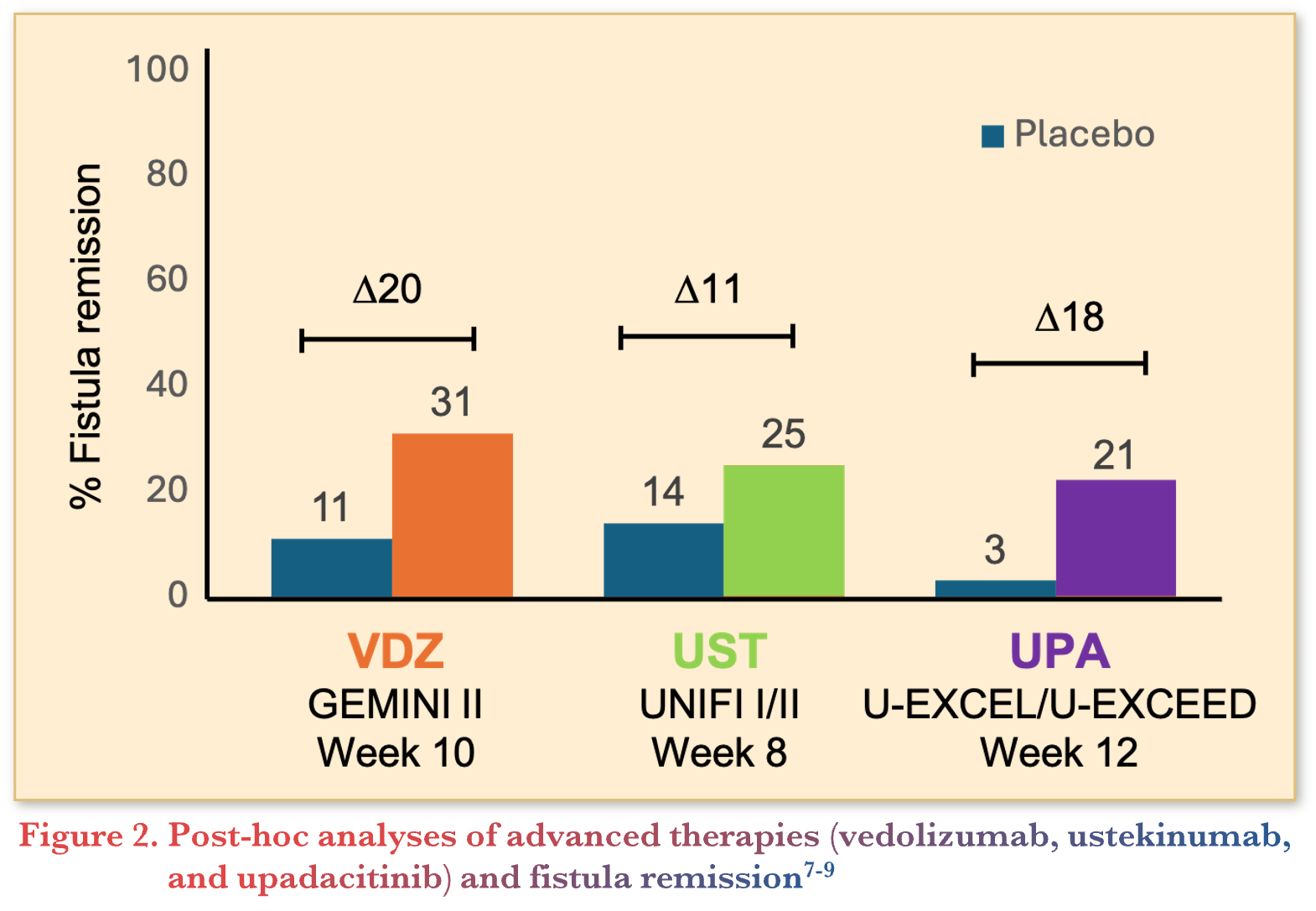
The DIVERGENCE-II trial evaluated filgotinib, a Janus Kinase (JAK) 1 inhibitor, in a dedicated pilot study for patients with PFCD.10 Patients treated with filgotinib at a dose of 200mg once daily achieved higher rates of combined clinical and radiologic fistula remission at 24 weeks compared with placebo (47.1% vs. 25%).10 These findings further support the use of JAK1 inhibitors for PFCD.
More recently, the USTAP randomized controlled trial, presented at the United European Gastroenterology Week (UEGW) in 2025, evaluated ustekinumab versus placebo in patients with active perianal fistulas, most of whom had failed anti-TNF therapy.11 At 12 weeks, ustekinumab demonstrated a favorable signal for combined remission compared with placebo, supporting efficacy of another non-anti-TNF therapy. Additionally, the FUZION trial of guselkumab, an anti–interleukin (IL)-23 therapy, in PFCD is underway and expected to report results within the next year.
It remains unknown which advanced therapy is the most effective treatment for PFCD. However, some studies suggest that there may be differences between therapies for preventing the development of fistulas in patients with CD. McCurdy and colleagues3 conducted a comparative cohort study using administrative data from a commercial database in the United States. Among patients who were naïve to advanced therapies, anti-TNF therapy was more effective than vedolizumab at preventing the development of luminal (HR = 0.66; 95% CI, 0.55–0.78; p <0.0001) and perianal fistulas (HR=0.88; 95% CI, 0.80–0.96; p=0.0045) and more effective than ustekinumab at preventing the development of luminal fistulas (HR=0.37; 95% CI, 0.30–0.46; p <0.0001).3 While these data suggest anti-TNF therapy could be considered a first-line therapy in patients who are at greater risk of developing fistulizing complications, future studies are needed to confirm these findings and to evaluate the comparative efficacy of anti-TNF therapy with newer advanced therapies such as IL-23 inhibitors and upadacitinib.
Controversy #5: Can Fistula Healing be Reliably Determined Clinically?
There are three potential outcomes of fistula healing as shown in Figure 3.12
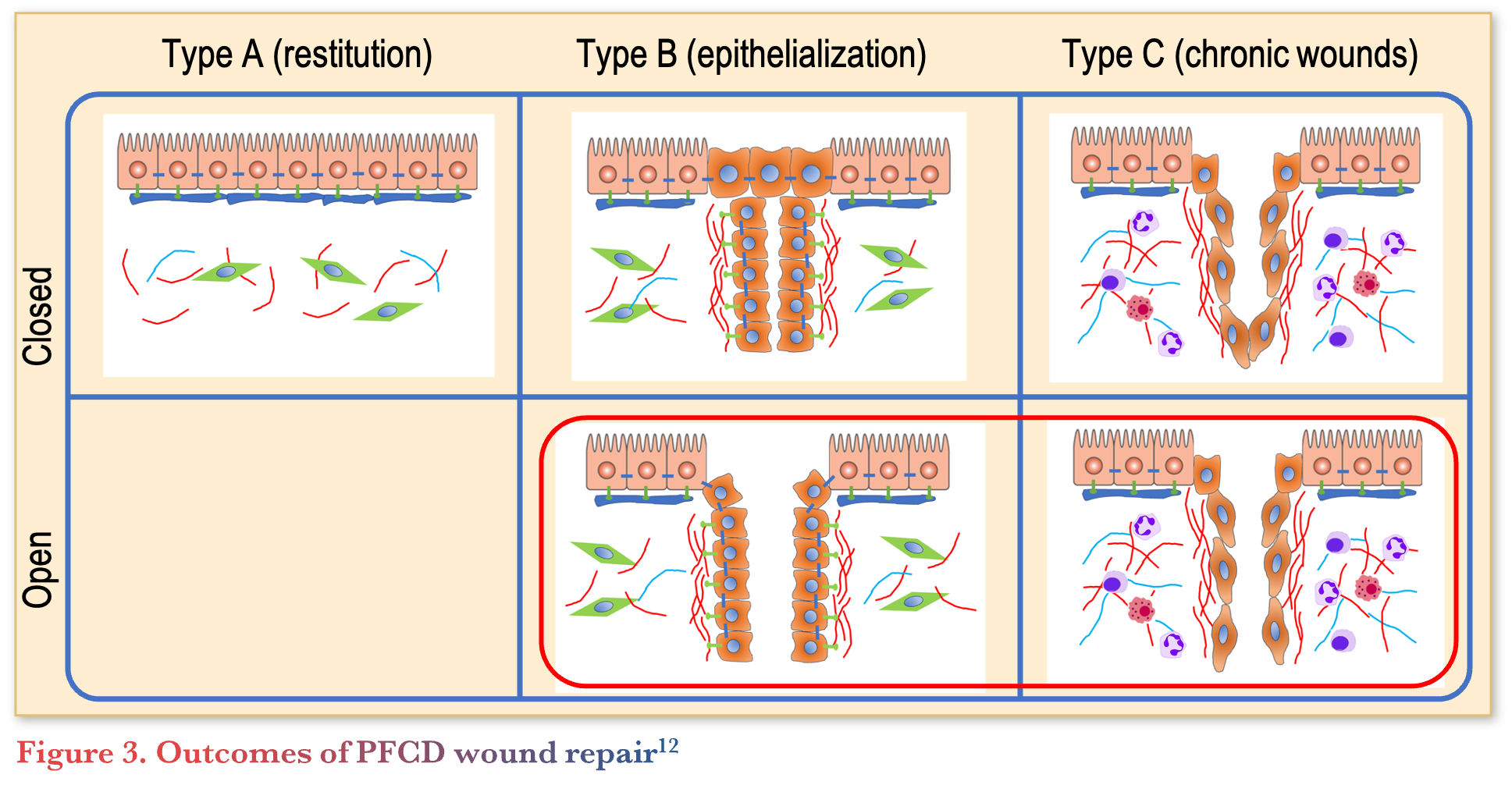
Type A healing consists of complete healing with an absence of residual fistula tracts, type B healing consists of epithelialized tracts without substantial inflammation and type C healing consists of chronic wounds with substantial inflammation. Type B and C healing can be difficult to distinguish clinically, since both classes may have open tracts associated with persistent fistula drainage. As a result, imaging techniques, such as pelvic MRI may be necessary to distinguish between classes, particularly in patients with residual symptoms. Differentiating between these classes is imperative as it may impact management. For instance, patients with persistent symptoms and substantial fistula tract inflammation (type C healing) require treatment strategies aimed at controlling inflammation, whereas patients without substantial fistula tract inflammation (type B healing) can be considered for surgical closure techniques.
Conclusion
PFCD remains a challenging manifestation of CD. Emerging evidence suggests that clinical examination alone may miss subclinical fistulas, and additional imaging may be warranted in selected high-risk patients. Diagnosing isolated PFCD in the absence of luminal inflammation can also be difficult, although tools such as VCE and emerging diagnostic criteria may help guide clinicians. While IFX remains the cornerstone of medical therapy, accumulating evidence suggests that other advanced therapies may also be effective. Finally, clinical assessment alone may not reliably determine fistula healing, and pelvic MRI can play an important role in distinguishing inflammatory from non-inflammatory fistula tracts and guiding management decisions.
Clinical Case
A 34-year-old male presents to the emergency department with recurrent perianal pain. On exam, there is a perianal abscess, confirmed by MRI of the pelvis (4 cm perianal abscess associated with a high transsphincteric fistula with multiple branches and hyperenhancement on T2 weighted imaging). The patient experienced a similar episode one year ago with complete resolution of symptoms after an incision and drainage procedure. An ileocolonoscopy at the time of the initial episode was normal including biopsies of the terminal ileum and colon.
Commentary
- The two most common etiologies for this clinical presentation include cryptoglandular fistulas and isolated PFCD. Less commonly, perianal fistulas can arise as a complication from hidradenitis suppurativa, tuberculosis or in rare cases, malignancy.
- It is critical to distinguish between CD-related fistulas from cryptoglandular fistulas because the management is different.
- Indicators of CD include complex fistula anatomy (greater involvement of the anal sphincter, branching fistulas, and multiple internal/external openings.
- In addition to ileocolonoscopy and cross-sectional imaging of the abdomen, VCE may detect occult luminal inflammation and thus help to discriminate cryptoglandular fistulas form PFCD.
- Fecal calprotectin may also help distinguish between cryptoglandular fistulas and PFCD.
Case Evolution
His fecal calprotectin is 350 µg/g, CTE is normal, and VCE demonstrates numerous aphthous ulcers in the third tertile of the small bowel suggesting a diagnosis of CD. Clinically, the abscess drains spontaneously and there is no longer perianal pain. The patient is afebrile and there is no longer an abscess clinically. However, the patient experiences daily fistula drainage requiring multiple changes of gauze throughout the day.
Commentary
- IFX is cornerstone medical therapy for PFCD and should be initiated once abscesses have been drained (spontaneously or surgically)
- Antibiotics should be combined initially with anti-TNF therapy.
- Immunomodulators, improve the pharmacokinetics of anti-TNF therapy and therefore may improve the efficacy of anti-TNF therapy for PFCD
- Subcutaneous (SC) IFX may achieve higher serum trough concentrations compared with intravenous dosing. However, the two modes of administration have yet to be compared in PFCD.
- If anti-TNF therapy cannot be tolerated or is contraindicated, alternate advanced therapies may be effective.
- Antibiotics should be combined initially with anti-TNF therapy.
- The role of EUA and seton placement is evolving.
- Both should be considered for complex fistula anatomy when an abscess is present. In addition to surgical drainage by EUA, setons may also help to promote tract drainage.
- In the absence of an abscess, it is unclear if EUA and setons improve the effectiveness of IFX.
- Setons should not be considered a long-term solution when the intent of treatment is complete fistula remission as they promote tract epithelization and prevent fistula closure.
- Setons can be removed after the induction period with anti-TNF therapy.
- In selected patients when the intent of treatment is symptom control alone, particularly for patients who have failed an attempt at seton removal, setons can be left in situ long-term
- Both should be considered for complex fistula anatomy when an abscess is present. In addition to surgical drainage by EUA, setons may also help to promote tract drainage.
Case Evolution
The patient is treated with IFX 5 mg/kg induction, followed by 5 mg/kg every 8 weeks and methotrexate 15 mg orally weekly. After 3 months the patient continues to have perianal drainage daily. On exam, exudate can be expressed by gentle finger palpation. However, there is no evidence of an abscess. Digital rectal exam (DRE) demonstrates mild anal canal stenosis. IFX serum trough concentrations are 9 µg/mL and fecal calprotectin is <9 µg/g.
Despite escalation of IFX to 10mg/kg every 4 weeks and resultant IFX serum trough concentrations of 25 µg/mL, as well as serial dilation of the anal canal stricture, the patient continues to have perianal drainage after 3 additional months. The patient’s symptoms are bothersome and impacting his quality of life.
Commentary
- Patients who do not achieve fistula remission should be assessed for persistent abscesses, subtherapeutic serum concentrations of anti-TNF therapy, persistent tract inflammation and anatomical complications such as anal strictures.
- Higher serum concentrations of anti-TNF therapy (up to doses of >20 µg/mL) have been associated with greater rates of fistula remission. Therefore, dose optimization is a reasonable strategy for patients with subtherapeutic serum concentrations and evidence of ongoing fistula tract inflammation.
- Alternate advanced therapies can be considered for persistently inflamed fistula tracts despite therapeutic concentrations of anti-TNF therapy
- Surgical closure options such as advancement flaps or Ligation of Intersphincteric Fistula Tract (LIFT) procedures can be considered in carefully selected patients with favorable anatomy and well-controlled luminal disease.
- Additional salvage strategies may also include hyperbaric oxygen therapy. This has been studied in case series and appears beneficial, but evidence remains limited.
- Fecal diversion is also a potential option and should be considered when perianal sepsis cannot be controlled or symptoms have a prominent impact on quality of life.
References
- Hwa Kim PH, Park SH, Jin K, et al. Supplementary anal imaging by magnetic resonance enterography in patients with crohn’s disease not suspected of having perianal fistulas. Clin Gastroenterol Hepatol. 2020;18(2):415–23.e4.
- Antaya M, Hudson AS, Lerner EP, et al. The impact of integrating pelvic magnetic resonance imaging at diagnosis on early detection of perianal Crohn’s disease in pediatrics. Am J Gastroenterol. doi:10.14309/ajg.0000000000003733.
- McCurdy JD, Stwalley D, Olsen MA, Deepak P. Comparative effectiveness of biologic therapies in preventing penetrating complications in patients with Crohn’s disease. Clin Gastroenterol Hepatol. 2024;22(2):377–85.e5.
- Hanna LN, Munster LJ, Joshi S, et al. Isolated perianal Crohn’s disease: a systematic review and expert consensus proposing novel diagnostic criteria and management advice. Lancet Gastroenterol Hepatol. 2025;10(8):757–68.
- Fung M, Farbod Y, Kankouni H, et al. Does combined medical and surgical treatment improve perianal fistula outcomes in patients with Crohn’s disease? A systematic review and meta-analysis. J Crohns Colitis. 2024;18(8):1261–9.
- McCurdy J, Munir J, Parlow S, et al. The impact of setons on perianal fistula outcomes in patients with Crohn’s disease treated with anti-TNF therapy: a multicentre study. Aliment Pharmacol Ther. 2025;61(10):1671–9.
- Sands BE, Gasink CR, Jacobstein DA, et al. Fistula healing in pivotal studies of ustekinumab in Crohn’s disease. Gastroenterology. 2017;152(5):S185.
- Sandborn WJ, Feagan BG, Rutgeerts P, et al; GEMINI 2 Study Group. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N Engl J Med. 2013;369(8):711–21.
- Colombel JF, Lacerda AP, Irving PM, et al. Efficacy and safety of upadacitinib for perianal fistulizing Crohn’s disease: a post hoc analysis of 3 phase 3 trials. Clin Gastroenterol Hepatol. 2025;23(6):1019–29.
- Reinisch W, Colombel JF, D’Haens GR, et al. Efficacy and safety of filgotinib for the treatment of perianal fistulising Crohn’s disease [DIVERGENCE 2]: a phase 2, randomised, placebo-controlled trial. J Crohns Colitis. 2024;18(6):864–74.
- Wils P, et al. Ustekinumab for fistulizing perianal Crohn’s disease: Week-12 results from the USTAP randomized placebo-controlled GETAID trial. Presented at: United European Gastroenterology Week (UEGW); October 6, 2025; Berlin, Germany.
- McCurdy JD, Hartley I, Behrenbruch C, et al. Management of perianal fistulizing Crohn’s disease according to principles of wound repair. Aliment Pharmacol Ther. 2025;61(4):600–13.
- Yarur AJ, Bruss A, Moosreiner A, et al. Higher intra-abdominal visceral adipose tissue mass is associated with lower rates of clinical and endoscopic remission in patients with inflammatory bowel diseases initiating biologic therapy: results of the constellation study. Gastroenterology. 2023;165(4):963–75.e5.
- Dewint P, Hansen BE, Verhey E, et al. Adalimumab combined with ciprofloxacin is superior to adalimumab monotherapy in perianal fistula closure in Crohn’s disease: a randomised, double-blind, placebo controlled trial (ADAFI). Gut. 2014;63(2):292–9.
- Meima-van Praag EM, van Rijn KL, Wasmann KATGM, et al. Short-term anti-TNF therapy with surgical closure versus anti-TNF therapy in the treatment of perianal fistulas in Crohn’s disease (PISA-II): a patient preference randomised trial. Lancet Gastroenterol Hepatol. 2022;7(7):617–26.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC CAGF AGAF Professor,
Department of Medicine Director, Division of Gastroenterology
McMaster University
Hamilton, ON
Contributing Author
Jeffrey McCurdy, MD PhD FRCPC
Assistant Professor of Medicine
University of Ottawa
Ottawa, ON
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
Eytan Wine, MD PhD FRCPC, University of Toronto, Toronto, ON
IBD Dialogue 2026·Volume 22 is made possible by unrestricted educational
grants from…
![]()
![]()









Published by Published by Kalendar Inc.., 7 Haddon Avenue,
Scarboro, ON M1N 2K7</span >
(c) Kalendar Inc. 2026. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not
necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.
