Many New Choices, But Who’s on First?—The Evolution of IBD Therapy
Many New Choices, But Who’s on First?—The Evolution of IBD Therapy
March 17, 2020
Issue 02
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by the contributing editor, Dr Brian Feagan, at the 20th annual national meeting, Mentoring in IBD XX: The Master Class, held November 15, 2019 in Toronto, Ontario.
Introduction
Biologic therapy, starting with the introduction of specific antagonists to tumor necrosis factor alpha (TNFα), and advanced by the addition of several new drug classes, has greatly improved the management of IBD. Multiple biologics are now available to treat ulcerative colitis (UC) and Crohn’s disease (CD); this raises critical questions regarding the relative therapeutic index of these agents and their positioning in treatment algorithms. In this review, both safety and efficacy will be discussed for the TNF antagonists, the anti-integrin vedolizumab, and IL-12/23 antagonists.
This discussion needs to be grounded in the context of several general observations. First, a large unmet medical need still exists with respect to efficacy. Early initiation of biologics in high-risk patients is required to achieve optimal benefits. Nevertheless, even when this paradigm is followed, remission rates remain less than ideal. Second, safety is a very important consideration for patients and clinicians. The “second generation” monoclonal antibodies vedolizumab and ustekinumab have infection rates that compare favourably to placebo, which is a clear differentiator from the TNF antagonists. Third, payors are under enormous financial pressure from the high cost of biologic drugs. Biosimilar TNF antagonists have entered the marketplace amid several controversies regarding their role in management.
In this milieu, professional societies have developed treatment guidelines that are based upon the best evidence available. The current Canadian Association of Gastroenterology (CAG) guidelines for treatment of UC and CD will be reviewed and commented upon in the light of relevant new data from important trials, including VARSITY and SERENE. Practical recommendations regarding choice of treatment will be offered. Finally, some speculations regarding the future of IBD management will be made.
Current Treatment Concepts in UC and CD
Management algorithms for both UC and CD have historically followed a step-care approach. This involved starting with less potent therapies (e.g., aminosalicylates), followed by corticosteroids if the initial therapy was not sufficient, followed by more potent therapies like biologics.(1) In UC, this largely continues to make sense; you can move up the step-care ladder and salvage patients who have not done well with the less potent bottom steps of the pyramid.
In CD, however, the concept of step care is flawed.(1) Due to the progressive, inflammation-driven nature of CD, intervening with highly active therapies earlier in the disease is preferred, particularly with patients who are deemed to be at high risk of progression. This approach is designed to dampen or eliminate inflammation, which reduces the long-term risk of irreversible damage and complications such as stricture, fistula/abscess and need for surgery.(1)
Overview of First-line Therapeutics for CD
In CD, first-line, highly active therapies include the three aforementioned classes of biologic agents: TNF antagonists, anti-integrins, and IL-12/23 antagonists. The following section provides a review of their mechanisms of action and evidence of efficacy and safety from the pivotal clinical trials.
TNF antagonists
The exact mechanism(s) of action of TNF antagonists are still not known, despite more than 20 years of experience with this class of biologics.(2) Basic science has shown us that these agents have multiple cellular effects, including cell-mediated toxicity, reverse signalling and induction of tolerance, but the therapeutic mechanisms of action are still not well understood.(2) What we do know is that neutralizing TNF has a rapid, powerful effect.
One aspect of the effect of TNF blockade that may be underappreciated is its effect on mood. The perceived rapidity of onset, with improvements reported very soon after starting TNF antagonist therapy is more likely linked to a central effect than any improvement in bowel symptoms.(3) Indeed, a randomized study using infliximab to treat depression found that among the subgroup of patients with baseline inflammatory biomarkers (baseline hs-CRP >5 mg/L), infliximab demonstrated an association with improved depressive symptoms compared to placebo.(3)
In terms of efficacy, while TNF antagonists truly were a game changer for IBD therapy, they are far from perfect. Remission rates in the maintenance period of the pivotal trials (e.g., CHARM with adalimumab)(4) are in the range of 30% to 40%, leaving considerable room for improvement. Even in combination with a thiopurine for early disease (e.g., infliximab + azathioprine in the SONIC study), the week 26 remission rate was only 57% for the combination group.(5)
The benefits of TNF antagonist therapy have gone beyond demonstrating improvements in clinical remission. Improvements in harder endpoints, like time to first surgery, hospitalization or complication in CD; or time to colectomy in UC, have also been demonstrated in clinical trials compared to conventional therapy.(6,7) These results suggest that these therapies can actually change the overall trajectory of the disease.
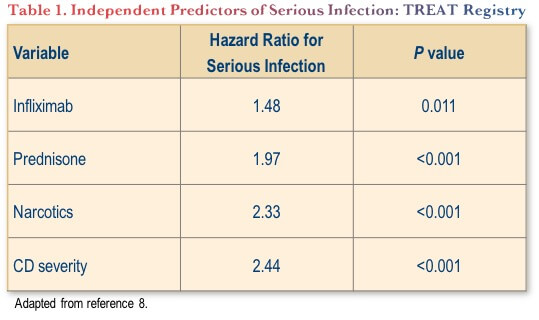
Where the TNF antagonists most clearly fall short of other highly active options in the current IBD therapeutic environment is in their safety profiles. Serious infection rates in particular are significantly increased with TNF antagonists. For example, in the large TREAT registry, infliximab treatment was independently associated with an approximate 48% increased risk of serious infection (Table 1).(8) This safety concern has not been replicated with later generation biologics for IBD, as discussed below.
Vedolizumab
The α4β7-integrin inhibitor vedolizumab selectively influences inflammation in the gut through interference with leukocyte trafficking, without the central effects that we see with TNF antagonists.(9)
The pivotal clinical trials in both UC and CD (GEMINI-1 and -2) showed a favourable efficacy for induction and maintenance, although there was a suggestion in the CD trial that vedolizumab did not work as quickly as had previously been seen with TNF antagonists.(10,11) More recently, there has been a head-to-head study comparing vedolizumab to adalimumab in UC that has refuted this.(12) In the VARSITY study, kinetics of response did not demonstrate any differences between vedolizumab and adalimumab in the early weeks of the study. After week 6, the response curves did begin to separate, becoming statistically significant in favour of vedolizumab by week 14.(12) By week 52, the primary endpoint of clinical remission was met by 31.3% in the vedolizumab group and 22.5% of the adalimumab group (absolute difference 8.8%; P=0.006).(12) There was also a significantly higher rate of mucosal healing at week 52 (39.7% vs. 27.7%; P<0.001). Although there was a nonsignificant trend in favour of adalimumab in this study with respect to corticosteroid-free remission, the reduction in corticosteroid exposure by mean dose was similar in both groups.(12)
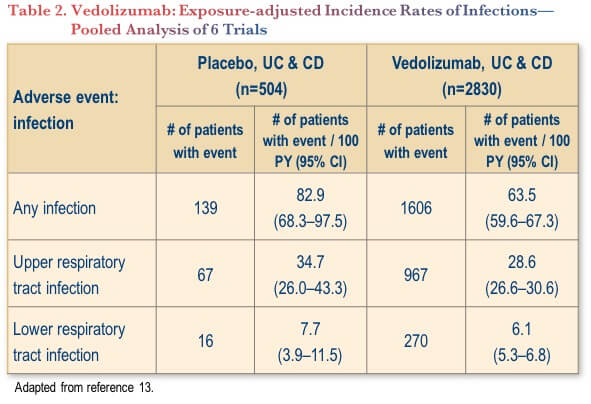
Vedolizumab’s safety profile has been investigated in a pooled analysis of six trials, involving 2830 patients (4811 patient-years of vedolizumab exposure).(13) The analysis showed that the incidence of infection (any grade) was actually significantly lower with vedolizumab compared to placebo (63.5 vs. 82.9 per 100 patient-years, with no overlapping of confidence intervals) (Table 2).(13) There were no significant differences between vedolizumab and placebo for upper or lower respiratory tract infections.(13)
Ustekinumab
Ustekinumab exerts its anti-inflammatory effects by binding to the p40 subunit common to IL-12 and IL-23. This inhibits signalling, cellular activation and downstream cytokine production through these pathways.(14-16)
The efficacy of this agent in both CD and UC has been shown in the UNITI and UNIFI studies, respectively,(17,18) with response and remission rates similar to the TNF antagonists or vedolizumab. Notably, these results were achieved despite the fact that many patients in these studies had already failed TNF antagonist and/or vedolizumab therapy.
Ustekinumab has also demonstrated a favourable safety profile across the IBD clinical trial experience to date, where infection rates have been similar to placebo.(17,18) Also notable in the context of ustekinumab is the long track record of use in psoriasis and psoriatic arthritis, where there no significant safety signals have been observed in post-marketing experience.(19)
Current Guidelines: Recommendations for Practice
Clinical practice guidelines, by their nature, lag behind the literature; the current Canadian guidelines for UC(20) and CD(21) are no exception. The VARSITY study(12) in particular, challenges the current UC guideline recommendation to consider TNF antagonists as the first biologic of choice. VARSITY provides a compelling argument to place vedolizumab as the first choice, with TNF antagonists reserved as second-line biologics.
Although there are no similar head-to-head biologic data in CD to make an efficacy-based recommendation for first-line therapy, the enhanced safety profiles of vedolizumab and ustekinumab might be enough to tip the scales in favour of these agents as preferred first-line biologics in CD, pushing TNF antagonists to 2nd-line status in CD as well.
Future Directions
The example of progressively better outcomes with subsequent generations of biologics has been clearly demonstrated in the psoriasis field, where the latest biologics (anti-IL-17 and anti-IL-23 agents) have been associated with remission rates of more than 90%.(22)
There have already been some results reported targeting the IL-23 pathway in CD. A phase 2 study evaluated risankizumab among 121 patients with CD, 93% of whom had previously failed at least one TNF antagonist.(23) At week 12, clinical remission rates were as high as 36.6% depending on dose and, importantly, endoscopic response was seen in up to 36.6% of patients as well. While these were positive results, it seems unlikely that the high remission rates seen in psoriasis will be achievable with single-agent therapy in IBD.
There are, however, other ways forward that may dramatically increase success rates with therapeutics in IBD. Precision medicine—matching individual patients to the best possible therapy using laboratory and/or molecular biomarkers—is under-developed, but holds promise for patients with IBD.
Combination therapy might also offer advantages over our current paradigms. Inflammation involves a complex interaction of biochemical processes and pathways. In theory, targeting two or more complementary pathways in this network has the potential to provide substantially greater benefit than one pathway alone. In theory, combinations that might have synergy include an anti-integrin + anti-IL-23; anti-integrin + JAK inhibitor; S1P-1 + anti-IL-23; and a locally acting TNF antagonist + anti-IL-23.
Finally, the role of the microbiome in IBD and its treatment is a rich area of research and there is hope that this will become an important component of integrated IBD management. Initial research with fecal microbiota transplantation has demonstrated promising results in terms of enhancing remission rates in UC.(24)
Conclusions
All three current classes of biologic used in IBD (anti-TNF, anti-integrin and anti-IL-12/23 have excellent therapeutic indices in comparison to conventional drugs (i.e., corticosteroids, small-molecule immunosuppressives). Vedolizumab and ustekinumab have safety profiles that are superior to TNF antagonists and should be viewed as first-line agents for both UC and CD. Future directions include exploring combination strategies, identifying biomarkers to use in precision medicine and incorporating modulation of the intestinal microbiome.
Clinical Case
Mary is a 28-year-old female working as an elementary school teacher. She has a 12-year history of left-sided ulcerative colitis, which has been well controlled on azathioprine. For two months she has had urgency, bloody diarrhea with four to six bowel movements (BMs) per day and abdominal cramps, mainly with her BMs. Because she wished to get pregnant, she recently stopped smoking. She was seen in your office one month ago, and you added both 5-ASA foam and budesonide MMX to her treatment. There has been no improvement in her symptoms.
Her investigations revealed:
- WBC: 8.0 x 109/L
- Hemoglobin: 109 g/L
- CRP: 22 mg/L
- Albumin: 38 g/L
- Fecal calprotectin: 1800 mcg/g
- Stool C and S negative
- C. difficile negative
- Colonoscopy showed a Mayo 2 pancolitis.
Commentary
Canadian clinical practice guidelines state that “In patients with moderate to severe active UC, we recommend oral corticosteroids as first-line therapy to induce complete remission. GRADE: Strong recommendation, moderate-quality evidence.”(20) One might interpret this to mean that a potent oral steroid (e.g., prednisone 40–60 mg / day) should be used, but in this case, the patient has already failed the oral budesonide MMX.
As such, since she has failed 5-ASA, a thiopurine and corticosteroids, one might consider using an advanced therapy.(20) The statement from the 2015 consensus paper reads: “In patients with UC who fail to respond to thiopurines or corticosteroids, we recommend anti-TNF therapy to induce complete corticosteroid-free remission. GRADE: Strong recommendation, high-quality evidence.”(20)
Since the time that the 2015 paper was prepared, there has been considerable evolution of the evidence base for advanced therapies in UC:
- There is further evidence with vedolizumab available, including a substantial expansion of real-life experience.(12,25,26)
- The oral small molecule tofacitinib has also become available for use in UC in Canada,(27) based largely on the results of the OCTAVE trials.(28)
- Ustekinumab was recently approved for UC in Canada,(29) based largely on evidence vs. placebo in the phase 3 UNIFI clinical trial.(18) Ustekinumab had already approved for CD.(29).
For Mary, a 28-year-old woman, it should be considered that tofacitinib is contraindicated in pregnancy.(27) This may not be the best choice for her. Further, since ustekinumab is not yet available for UC, if an advanced therapy is desired, the choice boils down to an anti-TNF agent or vedolizumab.
Another factor to consider is whether or not to continue the conventional immunomodulator (i.e., azathioprine) in combination with an anti-TNF agent. The UC SUCCESS study showed that patients receiving the combination of azathioprine and infliximab were more likely to achieve corticosteroid-free remission at 16 weeks than those receiving either monotherapy.(30)
Case Evolution
After discussion with the patient you decide to avoid prednisone and will start a biologic agent. Your experience has been that, with the assistance of patient support programs, all advanced therapies can be started within two weeks.
Commentary
There are three different anti-TNF agents available in Canada for the treatment of UC: infliximab, adalimumab and golimumab.
Anti-TNF agents have proven efficacy in clinical trials.(31–34)
In the ACT-1 study, week 8 remission rates were 39% in the infliximab 5 mg/kg treatment arm (n=121) and 15% in the placebo arm (P<0.001).(31) Intravenous infliximab infusion has a particularly rapid onset of action,(35) which may be desirable for Mary’s case.
In the ULTRA-1 study, among 186 patients with moderately to severely active UC, induction therapy with adalimumab led to an 18.5% remission rate at week 8, which was superior to the 9.2% rate observed with placebo (P=0.031).(32) In ULTRA-2 (n=494), 16.5% of adalimumab-treated patients achieved remission, compared to 9.3% of those in the placebo group (P=0.019).(33)
In the PURSUIT study (n=1064), remission rates at week 6 were 18% for golimumab and 6% for placebo (P<0.001).(34)
Anti-TNF therapies also have a robust track record of safety, including safety during pregnancy and lactation.(36)
The efficacy of vedolizumab in UC was established by the GEMINI-1 study.(10) Clinical remission rates at week 6 in that study were 16.9% and 5.4% among patients in the vedolizumab group and placebo group, respectively (P=0.001).(10)
More recently, vedolizumab was directly compared to adalimumab in the head-to-head VARSITY study.(12) At week 52, clinical remission was observed in a higher percentage of patients in the vedolizumab group than in the adalimumab group (31.3% vs. 22.5%; P=0.006) Endoscopic improvement rates were also significantly better with vedolizumab (39.7% vs. 27.7%; P<0.001). There was no significant difference in corticosteroid-free clinical remission rates, but the trend in this endpoint numerically favored adalimumab (21.8%) compared to vedolizumab (12.6%).
Exposure-adjusted incidence rates of infection were 23.4 and 34.6 events per 100 patient-years with vedolizumab and adalimumab, respectively. Exposure-adjusted incidence rates of serious infection were 1.6 and 2.2 events per 100 patient-years with vedolizumab and adalimumab, respectively.
In UC, the UNIFI study showed that i.v. ustekinumab was associated with a significantly higher rate of clinical remission at week 8 (15.6% for 130 mg dose or 15.5% for the 6 mg/kg dose) compared to placebo (5.3%; P<0.001 for both comparisons).(18)
Case Evolution
You decide on infliximab and continue azathioprine for now. She has done some research on her own and wishes to receive the “original” infliximab.
Commentary
With respect to biosimilars, there is no randomized controlled trial (RCT) available for UC. However, there was an RCT comparing original infliximab (Remicade®) with one of the biosimilars (CT-P13 [Inflectra®]) in CD, which found no significant difference in efficacy or safety between the two products.(37,38)
There is also substantial observational evidence in CD patients that there is no difference between these products in real-world use. This includes a French comparative equivalence cohort study of 5050 previously infliximab-naïve patients in the French nationwide health administrative database.(39) For the primary outcome (composite end point of death, CD-related surgery, all-cause hospitalization, and reimbursement of another biologic therapy), there was no significant difference between CT-P13 and originator infliximab.
With respect to therapeutic drug monitoring (TDM), there is a recent consensus paper from an international expert group (including three Canadian gastroenterologists) with clear recommendations. These differ by the type of biologic (anti-TNF or vedolizumab).(40)
For anti-TNF:
- It is appropriate to order drug/antibody concentration testing in responders at the end of induction for all anti-TNFs;
- It is appropriate to order drug/antibody concentration testing at least once during maintenance for patients on all anti-TNFs;
- It is appropriate to order drug/antibody concentration testing of anti-TNFs at the end of induction in primary non-responders; and
- It is appropriate to order drug/antibody concentration testing for all anti-TNFs in patients with confirmed secondary loss of response.
For vedolizumab:
- It is appropriate to order drug/antibody concentration testing for vedolizumab in responders at the end of induction;
- It is appropriate to order drug/antibody concentration testing at least once during maintenance for patients on vedolizumab;
- It is appropriate to order drug/antibody concentration testing for vedolizumab in non-responders at the end of induction; and
- It is appropriate to order drug/antibody concentration testing for vedolizumab in patients with confirmed secondary loss of response.
For the assessment of mucosal healing, there is no specific recommendation for follow-up assessment in the 2015 Canadian guidelines.(20)
Guidance from the STRIDE group states that mucosal healing should be a treatment goal in UC and it should be assessed by colonoscopy or flexible sigmoidoscopy every three months during the active phase.(41)
Case Evolution
She receives a standard three-dose induction regimen of infliximab, which leads to complete clinical remission. She remains on 5 mg/kg every eight weeks. At her six-month follow-up visit she expresses concern about being on combination therapy and is particularly concerned about the safety of azathioprine. You agree to stop azathioprine. Six months later she experiences a relapse, with four to six bloody BMs/day, CRP 15 mg/L and fecal calprotectin 800 mcg/g. A serum trough level of infliximab is 14 mcg/mL. She has heard that there is a new oral agent available for treating UC.
Commentary
There are a few alternatives to consider here:
- Increase the dose of infliximab
- Ask her to consider adding azathioprine back in
- Switch to another anti-TNF (adalimumab or golimumab)
- Switch to vedolizumab
- Switch to tofacitinib
Increasing to a supra-therapeutic dose of infliximab is an option, but is probably not the best one considering the other options. She also probably would not agree to returning to combination therapy with azathioprine.
For efficacy failures, switching class is usually favored when available, rather than switching within the class, so adalimumab or golimumab are also reasonable, but probably not the best options.
Tofacitinib, as mentioned above, is contraindicated in pregnancy and lactation,(27) so likely not the best choice for our 28-year-old female patient. There have also been safety concerns raised with the 10 mg dose of this molecule in the rheumatoid arthritis indication, where there have been post-marketing reports of treatment-emergent pulmonary embolism.(42)
Vedolizumab appears to be the best treatment for this patient at this time.
Case Evolution
After some discussion you decide to switch her to vedolizumab. She receives her induction doses at weeks 0, 2 and 6. You see her just before her first maintenance therapy 8 weeks later. She has had a partial clinical response but still has 3 to 4 loose BMs/day, half are bloody with urgency, CRP 10 and fecal calprotectin 500. Vedolizumab TDM is now available in Canada. Her trough level is <3.5 mcg/mL.
Commentary
As mentioned above, a recent consensus paper made the following recommendations for TDM with vedolizumab:(40)
- It is appropriate to order drug/antibody concentration testing for vedolizumab in responders at the end of induction;
- It is appropriate to order drug/antibody concentration testing at least once during maintenance for patients on vedolizumab;
- It is appropriate to order drug/antibody concentration testing for vedolizumab in non-responders at the end of induction; and
- It is appropriate to order drug/antibody concentration testing for vedolizumab in patients with confirmed secondary loss of response.
This paper stated there is currently insufficient evidence to define thresholds for action with respect to TDM results.
However, a recent meta-analysis reported that patients with vedolizumab trough concentration >20 mcg/mL at week 6, and >12 mcg/mL during maintenance may be associated with better outcomes.(43) As such, Mary’s current 3.5 mcg/mL result seems to be too low—dose optimization may be appropriate here.
Case Evolution
Her vedolizumab dose is escalated to 300 mg q4 weeks. Her symptoms gradually improve leading to clinical remission. She is maintained on 300 mg IV q4 weeks.
References
-
Khanna R, Seow C. Biomarkers in the Management of Crohn’s Disease. 2018;154:2270–2.
-
Olesen CM, Coskun M, Peyrin-Biroulet L, et al. Mechanisms behind efficacy of tumor necrosis factor inhibitors in inflammatory bowel diseases. Pharmacol Ther. 2016;159:110–9.
-
Raison CL, Rutherford RE, Woolwine BJ, et al. A randomized controlled trial of the tumor necrosis factor antagonist infliximab for treatment-resistant depression: the role of baseline inflammatory biomarkers. JAMA Psychiatry. 2013;70(1):31–41.
-
Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: the CHARM trial. Gastroenterology. 2007;132(1):52–
-
Colombel JF, Sandborn WJ, Reinisch W, et al. Infliximab, azathioprine, or combination therapy for Crohn’s disease. N Engl J Med. 2010;362(15):1383–95.
-
Khanna R, Bressler B, Levesque BG, et al. Early combined immunosuppression for the management of Crohn’s disease (REACT): a cluster randomised controlled trial. Lancet. 2015;386(10006):1825–34.
-
Sandborn WJ, Rutgeerts P, Feagan BG, et al. Colectomy rate comparison after treatment of ulcerative colitis with placebo or infliximab. Gastroenterology. 2009;137(4):1250–60.
-
Lichtenstein G, Cohen RD, Feagan BG, et al. Safety of Infliximab and Other Crohn’s Disease Therapies: TREAT™ Registry Data With 27, 762 Patient-Years of Follow-up [AGA abstract T1040]. Gastroenterology. 2010;138(5 Suppl.1):S-475.
-
Mosli MH, Rivera-Nieves J, Feagan BG. T-cell trafficking and anti-adhesion strategies in inflammatory bowel disease: current and future prospects. Drugs. 2014;74(3):297–311.
-
Feagan BG, Rutgeerts P, Sands BE, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013;369(8):699–710.
-
Sandborn WJ, Feagan BG, Rutgeerts P, et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N Engl J Med. 2013;369(8):711–21.
-
Sands BE, Peyrin-Biroulet L, Loftus EV Jr, et al. Vedolizumab versus Adalimumab for Moderate-to-Severe Ulcerative Colitis. N Engl J Med. 2019;381(13):1215–26.
-
Colombel JF, Sands BE, Rutgeerts P, et al. The safety of vedolizumab for ulcerative colitis and Crohn’s disease. Gut. 2017;66(5):839–51.
-
Sandborn W, Gasink C, Blank M, et al. A multicenter, double-blind, placebo-controlled phase 3 study of ustekinumab, a human IL-12/23P40 mAB, in moderate-severe Crohn’s disease refractory to anti-TFNα: UNITI-1. Presented at CCFA 2015; Abstract O-001.
-
Rutgeerts P, Gasink C, Blank M, et al. A multicentre, double-blind, placebo-controlled phase 3 study of ustekinumab, a human interleukins-12/23p40 mab, in moderate-severe Crohn’s disease refractory to anti-tumour necrosis factor α: UNITI-1 [ECCO abstract #OP014]. J Crohn’s Colitis. 2016;10(Suppl.1):S11–S12.
-
Sandborn W, Feagan BG, Gasink C, et al. A Phase 3 Randomized, Multicenter, Double-Blind, Placebo-Controlled Study of Ustekinumab Maintenance Therapy in Moderate – Severe Crohn’s Disease Patients: Results From IM-UNITI [AGA abstract #768]. Gastroenterology. 2016; 150(4 Suppl. 1):S157–S158.
-
Feagan BG, Sandborn WJ, Gasink C, et al. Ustekinumab as Induction and Maintenance Therapy for Crohn’s Disease. N Engl J Med. 2016;375(20):1946–60.
-
Sands BE, Sandborn WJ, Panaccione R, et al. Ustekinumab as Induction and Maintenance Therapy for Ulcerative Colitis. N Engl J Med. 2019;381(13):1201–14.
-
Papp K, Gottlieb AB, Naldi L, et al. Safety Surveillance for Ustekinumab and Other Psoriasis Treatments From the Psoriasis Longitudinal Assessment and Registry (PSOLAR). J Drugs Dermatol. 2015;14(7):706–14.
-
Bressler B, Marshall JK, Bernstein CN, et al. Clinical practice guidelines for the medical management of nonhospitalized ulcerative colitis: the Toronto consensus. Gastroenterology. 2015;148(5):1035–58.
-
Panaccione R, Steinhart AH, Bressler B, et al. Canadian Association of Gastroenterology Clinical Practice Guideline for the Management of Luminal Crohn’s Disease. Clin Gastroenterol Hepatol. 2019;17(9):1680–713.
-
Sawyer LM, Malottki K, Sabry-Grant C, et al. Assessing the relative efficacy of interleukin-17 and interleukin-23 targeted treatments for moderate-to-severe plaque psoriasis: A systematic review and network meta-analysis of PASI response. PLoS One. 2019;14(8):e0220868.
-
Feagan BG, Sandborn WJ, D’Haens G, et al. Induction therapy with the selective interleukin-23 inhibitor risankizumab in patients with moderate-to-severe Crohn’s disease: a randomised, double-blind, placebo-controlled phase 2 study. Lancet. 2017;389(10080):1699–709.
-
Costello SP, Hughes PA, Waters O, et al. Effect of Fecal Microbiota Transplantation on 8-Week Remission in Patients With Ulcerative Colitis: A Randomized Clinical Trial. 2019;321(2):156–164.
-
Amiot A, Serrero M, Peyrin-Biroulet L, et al. Three-year effectiveness and safety of vedolizumab therapy for inflammatory bowel disease: a prospective multi-centre cohort study. Aliment Pharmacol Ther. 2019;50(1):40–53.
-
Chaparro M, Garre A, Ricart E, et al. Short and long-term effectiveness and safety of vedolizumab in inflammatory bowel disease: results from the ENEIDA registry. Aliment Pharmacol Ther. 2018; 48(8):839–51.
-
Pfizer Canada ULC. PrXeljanz® Product Monograph. Date of Preparation: July 2, 2019.
-
Sandborn WJ, Su C, Sands BE, et al. OCTAVE Induction 1, OCTAVE Induction 2, and OCTAVE Sustain Investigators. Tofacitinib as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2017;376: 1723–36.
-
Janssen Inc. PrStelara® Product Monograph. Date of Revision: January 23, 2020.
-
Panaccione R, Ghosh S, Middleton S, et al. Combination therapy with infliximab and azathioprine is superior to monotherapy with either agent in ulcerative colitis. Gastroenterology. 2014;146(2):392–400.
-
Rutgeerts P, Sandborn WJ, Feagan BG, et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2005;353:2462–76.
-
Reinisch W, Sandborn WJ, Hommes DW, et al. Adalimumab for induction of clinical remission in moderately to severely active ulcerative colitis: results of a randomised controlled trial. Gut. 2011;60:780–7.
-
Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients with moderate-to-severe ulcerative colitis. Gastroenterology. 2012;142:257–65.
-
Sandborn WJ, Feagan BG, Marano C, et al. Subcutaneous golimumab induces clinical response and remission in patients with moderate-to-severe ulcerative colitis. Gastroenterology. 2014;146:85–95.
-
Vasudevan A, Gibson PR, van Langenberg DR, et al. Time to clinical response and remission for therapeutics in inflammatory bowel diseases: What should the clinician expect, what should patients be told? World J Gastroenterol. 2017;23(35):6385–402.
-
Mahadevan U, Robinson C, Bernasko N, et al. Inflammatory Bowel Disease in Pregnancy Clinical Care Pathway: A Report From the American Gastroenterological Association IBD Parenthood Project Working Group. Gastroenterology. 2019;156(5):1508–24.
-
Kim YH, Ye BD, Pesegova M, et al. Phase III randomised, double-blind, controlled trial to compare biosimilar infliximab (CT-P13) with innovator infliximab in patients with active Crohn’s disease: early efficacy and safety results. Presented at ECCO 2017; Abstract #DOP061.
-
Ye BD, Kim YH, Pesegova M, et al. Phase III Randomized Controlled Trial to Compare Biosimilar Infliximab (CT-P13) with Innovator Infliximab in Patients with Active Crohn’s Disease: 1-Year Maintenance and Switching Results. Gastroenterology. 2018;154(6 Suppl.1):S167–8.
-
Meyer A, Rudant J, Drouin J, et al. Effectiveness and Safety of Reference Infliximab and Biosimilar in Crohn Disease: A French Equivalence Study. Ann Intern Med. 2019;170(2):99–107.
-
Papamichael K, Cheifetz AS, Melmed GY, et al. Appropriate Therapeutic Drug Monitoring of Biologic Agents for Patients With Inflammatory Bowel Diseases. Clin Gastroenterol Hepatol. 2019;17(9):1655–68.
-
Peyrin-Biroulet L, Sandborn W, Sands BE, et al. Selecting therapeutic targets in inflammatory bowel disease (STRIDE): determining therapeutic goals for treat-to-target. Am J Gastroenterol. 2015; 110(9):1324–38.
-
Health Canada. Clinical trial finds an increased risk of blood clots in the lungs and of death in rheumatoid arthritis patients taking high dose of tofacitinib (sold as Xeljanz or Xeljanz XR). Information Update #RA-69336; March 15, 2019.
-
Singh S, Dulai PS, Vande Casteele N, et al. Systematic review with meta-analysis: association between vedolizumab trough concentration and clinical outcomes in patients with inflammatory bowel diseases. Aliment Pharmacol Ther. 2019;50(8):848–57.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC AGAF, Director, Division of Gastroenterology, Professor, Department of Medicine, McMaster University, Hamilton, ON
Contributing Editor
Brian G. Feagan, MD FRCPC, Professor of Medicine, Epidemiology and Biostatistics, Senior Scientific Officer, Robarts Clinical Trials Inc., Robarts Research Institute, Western University, London, ON
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Anne M. Griffiths, MC FRCPC, University of Toronto, Toronto, ON
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
IBD Dialogue 2020·Volume 16 is made possible by unrestricted educational grants from…
![]()



Published by Catrile & Associates Ltd., 1B-391 Berkeley Street, Toronto, ON M5A 2X8
(c) Catrile & Associates Ltd., 2020. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.
