Nutrition Therapy for IBD: A Primer for Adult GI
Nutrition Therapy for IBD: A Primer for Adult GI
February 4, 2020
Issue 01
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by the contributing editor, Dr Eytan Wine, at the 20th annual national meeting, Mentoring in IBD XX: The Master Class, held November 15, 2019 in Toronto, Ontario.
Introduction
Questions and concerns about diet are extremely common among patients with IBD. Traditionally, research has not kept pace with the demand for answers regarding dietary management in IBD, but this appears to be changing.
Dietary management for patients with IBD can be separated into three components: assessment, supportive therapy, and nutritional therapy. The majority of this review will focus on the latter—diet as a primary therapy for IBD. There have been considerable advances in this area of research over the past several years; the key studies will be reviewed below.
However, this review will first touch on principles of nutritional support, providing some practical advice for communication with patients.
Nutritional Support for Patients with IBD
In a recent Canadian survey of IBD patients, caregivers and healthcare professionals, participants were asked to rank gaps in the field of IBD research. The top three priorities were found to be “What are the causes of IBD?”, “Can IBD be prevented?” and “What role does diet have in the management of IBD?”(1)
With respect to the third question, when clinicians are asked about treating IBD with diet, the pieces of advice most of us would probably give includes that: there is no specific dietary therapy for IBD; one should identify potential food triggers and avoid them; and following the Canada Food Guide is recommended.
The approach taken by the author and colleagues at the Edmonton Pediatric IBD clinic includes counselling about possible dietary contributors to IBD pathogenesis. There is a reduced risk of IBD among people with a diet rich in fruits and vegetables and a higher risk among those who consume diets high in animal fat, animal protein and refined sugar.(2) We also discuss food choices that may help improve IBD for people in remission, advising that they follow a Mediterranean-type diet instead of a typical Western diet.(3) We provide a handout with a column of good food choices and a column of foods to avoid (freely accessible at https://www.kidsibd.ca/nutrition).
Some of the practical tips that we also try to include in our patient education are shown in Table 1.
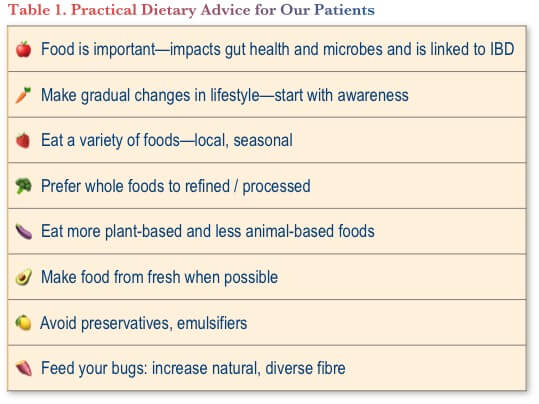
All that said, it is emphasized that people with IBD should consult their doctor and/or dietitian to discuss any particular restrictive or avoidance diet they are considering—to ensure adequate nutrition is still being provided. Importantly, while we have traditionally explained that there is no “IBD diet” that can cure a patient’s IBD, this is an evolving field with much promise for the future.
Diet as Primary Treatment for IBD
Whereas there is little evidence available to guide specific recommendations for supportive nutritional therapy for patients with IBD, there is a solid and growing body of evidence showing the value of nutritional therapy as a primary treatment.
EEN
The value of exclusive enteral nutrition (EEN) in pediatric Crohn disease (CD) is well established. The evidence clearly shows that six to eight weeks of liquid diet by mouth or nasogastric tube, with no other food, is safer and at least as effective as steroids for induction of remission.(4) Guidelines have recognized this by placing EEN as a preferred therapy for children with active luminal CD. For example, in this population, the ECCO/ESPGHAN guidelines state that “EEN is recommended as first line therapy to induce remission”.(5)
A meta-analysis of studies comparing EEN vs. glucocorticoids in pediatric populations showed that EEN was at least as effective as glucocorticoids for induction of remission (odds ratio [OR] 1.26, 95% confidence interval [CI] 0.77 to 2.05).(6) For mucosal healing, EEN was found to be superior to glucocorticoids (OR 4.5; 95% CI 1.46 to 12.23).(6) Further research published since the meta-analysis was conducted further support these findings. A small randomized, prospective trial (n=19) showed that after eight weeks of EEN or glucocorticoid therapy, the median CDEIS score was 1 (range 0–21) for EEN and 7 (3–14) for glucocorticoids (P<0.05).(7) Avoidance of steroid exposure in pediatric IBD is critical for growth and bone development.
Up until recently, the evidence for diet as a primary therapy for IBD in adults has been very weak. The British Society of Gastroenterology consensus guidelines for IBD in adults, for example, includes two statements about dietary therapy and both are classified as weak recommendations:(8)
- “. . . a low FODMAP diet may be used to treat functional bowel symptoms in IBD patients (weak recommendation, low quality evidence);” and
- “. . . EEN may be used to induce remission in mild to moderate CD patients where avoidance of corticosteroid is desired, and in those who are motivated to adhere strictly to EEN for up to 8 weeks (weak recommendation, very low-quality evidence)”.
While the evidence was deemed to be of low quality, there is indeed some evidence to support the use of EEN in adults. A controlled trial of 51 adult patients with IBD, published by Dr. Greenberg in 1988, showed that 21 days of total parenteral nutrition or EEN was associated with an enhanced remission rate in the short term among adults with CD, but did not impact the one-year remission rate.(9) EEN has also been shown to be beneficial among patients awaiting surgery for CD, where it has been associated with less need for surgery, reduced systemic inflammation, shorter operative times and a reduced incidence of post-operative abscess or anastomotic leak.(10) A single-centre review of 50 consecutive adult patients using EEN for CD showed that clinical and biochemical parameters improved compared to baseline.(11) Adherence to EEN in this cohort was 74%.
Adherence to EEN is the primary barrier to its use. An international survey of 146 participants from 26 countries (including Canada) showed that poor adherence to EEN related to its monotony was reported by 57% of Canadian respondents and 58% overall.(12) Poor adherence due to palatability was reported by 29% of Canadian respondents and 42% overall.(12)
Other dietary approaches
There are also dietary approaches other than EEN that have been studied for treatment of IBD, including partial enteral nutrition (PEN), specific carbohydrate diet (SCD), an individualized food-based diet (Crohn’s Disease Treatment with EATing [CD-TREAT]), and the Crohn’s disease exclusion diet (CDED). The commonality of all these approaches is food exclusion of one type or another.(13)
PEN is not recommended for induction. It has been shown to be significantly inferior to EEN, with a low remission rate (15% in one trial in children).(14)
Because EEN is thought to work through the elimination of potentially damaging foods,(15) a number of more targeted dietary approaches have sought to more specifically limit those components. The specific carbohydrate diet, which takes that approach, has been used in multiple studies in children with improvement of symptoms, but no documented impact on mucosal healing.(16,17)
The CD-TREAT dietary approach is a food-based diet designed to replicate the effects of EEN.(18) To date, it has been trialled in healthy volunteers and in a pilot trial in children with active CD. Its efficacy still needs to be assessed in large, well-controlled clinical trials. The hope is that this approach could be used interchangeably with EEN, and also as a long-term dietary maintenance therapy.(18)
The most recent evidence for dietary therapy in IBD comes from a prospective, randomized, controlled trial (RCT) of CDED in children.(19) This is a whole-food diet coupled with PEN, which is designed to reduce exposure to dietary components that have adverse effects on the microbiome and intestinal barrier. The CDED includes a list of mandatory, allowed and disallowed foods and products. Generally, it excludes gluten, gluten-free baked goods, yeast, dairy products, animal fat, processed meats and products containing emulsifiers; it is a low-sugar but not low-carbohydrate diet. The CDED approach contains at least 18–20 grams of fiber per day.(19)
In the RCT, 78 patients were initially included; four withdrew in the first 48 hours due to intolerance, leaving 74 for analysis. The subjects were randomized to the CDED + 50% PEN with ModulenTM for weeks 0–6, followed by 25% Modulen and the CDED step-down diet for weeks 6–12; or EEN with Modulen for weeks 0–6 followed by 25% Modulen + an unrestricted diet for weeks 6–12.
The primary and secondary outcome results are shown in Figure 1. For the primary outcome of dietary tolerance, the CDED approach was found to be significantly better than EEN; the combination of CDED and PEN was tolerated in 39/40 children (97.5%), whereas EEN was tolerated by 28/38 children (73.6%) (OR for tolerance of CDED and PEN, 13.92; 95% CI 1.68 to 115.14; P=0.002).(19)

Another important finding in favour of the CDED + PEN approach was a significant improvement in sustained remission at week 12, compared to EEN. Using the threshold of PCDAI ≤10, 75% of patients in the CDED group achieved sustained corticosteroid-free remission, compared to 45% of the EEN group (P<0.01).(15) For the threshold of PCDAI ≤7.5, the results were also significant: 65% for CDED and 38% for EEN (P<0.01).(19)
Analysis of the microbiome was conducted in 70 of the patients in this study. In the EEN group between week 6 and week 12, the composition of the microbiome (particularly Proteobacteria) tended to rebound to the baseline composition after returning to regular diet. This phenomenon was not observed in the CDED group, where positive changes were generally sustained. These observations suggest that the foods excluded in the CDED are likely driving the dysbiosis-led inflammation.
The CDED has also been evaluated in a small study (N=21) as induction therapy for adults (n=11) and children (n=10) who had failed remission with a biologic therapy.(20) Most of the patients (17/21) had failed combination therapy, 10 of 21 had failed two biologics and most (17/21) had undergone dose escalation. After six weeks of therapy with the CDED, 13/21 patients (62%) had achieved remission (Harvey-Bradshaw Index of 3 or lower).(20)
There are also ongoing studies underway evaluating the CDED, including a long-term international study involving approximately 70 patients (DietOmics Study)(21) and a 24-week study in adults.(22)
Conclusions
Patients with IBD (and parents of children with IBD) frequently have questions about appropriate diet. Although there remain large knowledge gaps in this field, there has been recent progress. For supportive diet therapy, nutritional advice is based on sound rationales, but lacks solid evidence. With respect to diet as primary treatment for IBD, both EEN and CDED have been shown to be effective for CD, although neither is perfect. Patients must understand that any prescribed diet can be difficult to implement and sustain, and that professional support is required.
Clinical Case
Emma is an 18-year-old female you meet as she transitions from pediatric to adult gastroenterology care. She attends with both parents.
You review her history. She was diagnosed at a pediatric hospital at age 17 with ileocecal disease after presenting with pain, anorexia, weight loss and iron deficiency. Magnetic resonance enterography (MRE) at diagnosis showed 35 cm of disease with early stricture but no features of obstruction. She was managed with exclusive enteral nutrition (EEN) for 12 weeks. She has continued to use EEN by nasogastric tube (with self-intubation) on and off when she starts to feel unwell. She is taking no other therapy. Emma and her parents have read about medications for IBD, but prefer to continue using EEN as needed.
Commentary
Current Canadian guidelines make the following recommendations for the management of active pediatric CD:(23)
- In patients with CD, we suggest exclusive enteral nutrition to induce clinical remission;
- In patients with CD, we recommend against partial enteral nutrition to induce clinical remission;
- In patients with mild to moderate active CD despite use of sulfasalazine, 5-ASA, oral budesonide, or exclusive enteral nutrition, we suggest oral prednisone to induce clinical remission; and
In patients with CD in remission, we suggest that if partial enteral nutrition is used it should be combined with other medications to maintain clinical remission. A meta-analysis published in 2017 compared the efficacy of EEN and corticosteroids for the induction of remission in pediatric CD.(6) There were eight studies identified (total N=451) that met inclusion criteria and had data that could be abstracted into the meta-analysis. EEN was found to be as effective as corticosteroids in inducing remission (Odds ratio [OR]=1.26, 95% CI 0.77 to 2.05). EEN and corticosteroid efficacy was comparable for both newly diagnosed (OR=1.61, 95% CI 0.87 to 2.98) or relapsed CD (OR=0.76, 95% CI 0.29 to 1.98]). Importantly, in this meta-analysis, intestinal healing was significantly more likely among patients receiving EEN vs. corticosteroids (OR=4.5, 95% CI 1.64 to 12.32]).
In adults, however, the data on EEN are limited, and is not widely used.(24) Nor is this approach endorsed by clinical practice guidelines.(25)
The use of PEN is appealing to many, as the monotony of exclusively drinking parenteral formula is often a reason for poor acceptance of / adherence to EEN.(26)
There has only been one study of PEN in active CD, and this study reported no effect of this approach on disease activity.(27)
There is, however, evidence of efficacy of PEN for maintenance, with a number of smaller studies reporting superior impact on relapse rates compared to normal diet.(28–33) This may be an option for Emma as maintenance.
In one of those studies, maintenance 6-mercaptopurine was an active control arm. Both PEN and 6-MP were superior to normal diet. Although there was no statistically significant difference between PEN and the medical therapy for relapse rates, 6-MP was numerically better (43% relapse rate for 6-MP at one year, 56% for PEN, 79% for normal diet).(28)
More recently, PEN has been evaluated in combination with a specific exclusion diet—the Crohn’s Disease Exclusion Diet (CDED)—for induction of remission in pediatric CD.(19) The diet is designed to reduce exposure to dietary components that have adverse effects on the microbiome and intestinal barrier. Seventy-eight children were randomly assigned to receive CDED plus 50% of calories from formula (Modulen, Nestlé) for 6 weeks (stage 1) followed by CDED with 25% PEN from weeks 7 to 12 (stage 2) (n=40) or EEN for 6 weeks followed by a free diet with 25% PEN from weeks 7 to 12 (n=38).
At week 12, 28 of 37 children (75.6%) given CDED plus PEN were in corticosteroid-free remission compared with 14 of 31 (45.1%) children given EEN and then PEN (P=0.01; OR 3.77, 95% CI 1.34 to 10.59). Corticosteroid-free remission was associated with sustained reductions in inflammation (based on serum level of C-reactive protein and fecal level of calprotectin) and fecal Proteobacteria. However, as CDED was proved effective in induction of remission, it may not apply to Emma’s case as a maintenance therapy.
Case Evolution
You present some potential options for Emma to consider, including PEN and medical maintenance therapy. Emma agrees to consider PEN and to do some more reading about medications before her next visit. You propose some additional investigation, to which she agrees. She asks if she needs to take any vitamins.
Commentary
Vitamin and mineral deficiencies are common among IBD patients and warrant supplementation to restore recommended values.(34) There is, however, a lack of evidence-based approaches supported by well-designed clinical trials to document the optimal supplementation level and to assess the benefits of supplementation exceeding the recommended daily allowance.(34)
Common deficiencies that may require supplementation include calcium, vitamin D, folic acid, iron, vitamin B12, vitamins A, E and K, and zinc.(35) Recommending a multivitamin, together with vitamin D and calcium supplementation, is a reasonable approach.(36) Iron and vitamin B12 should be monitored and, if appropriate, supplementation provided.(36) In this case, iron is low and supplementation should be recommended.
It does not appear that supplementation with fish oil has any substantial impact on IBD outcomes, and there are potential concerns with tolerability (increased risk of diarrhea and other GI symptoms).(37)
Case Evolution
Emma is keen to take everything you suggest as supplements. She completes her investigation and returns for her 3-month follow-up.
- Fecal calprotectin: 276 mcg/g
- C-reactive protein (CRP): 3.5 mg/L
- Hemoglobin (Hb) 115 g/L with mean corpuscular volume (MCV) 81 fL
- Colonoscopy: Aphthous ulceration in cecum and ileum. Distorted ileocecal valve.
- MRE: No change from diagnosis except subtle dilation of the lumen above the stricture.
You suggest that Emma start a biologic therapy. Emma has read about available therapies and is worried about side effects. She wants to restart tube feeds and will not start other therapies.
Commentary
Guidelines are clear that this patient should be receiving systemic therapy for her active disease. Her younger age and disease features put her at increased risk of poor prognosis and relapse. As such, it is reasonable to recommend a biologic therapy with or without concomitant thiopurine (or methotrexate with or without oral corticosteroid is the other guideline-recommended therapy for this scenario).(25)
Counseling should include a frank discussion of benefits and risks of treating vs. not treating her CD with systemic medication. Also, the benefits and risks of the various potential therapies need to be part of the discussion.
If she is set on not taking any medication, a referral to a dietitian may be helpful.
Case Evolution
Emma met a friend on a chat forum who has CD and does nutrition therapy without the tube. She now wants to resume exclusive enteral nutrition, but drinking the formula rather than using a tube.
Commentary
There are no head-to-head studies available comparing any of the enteral nutrition formulas for patients with IBD. However, the study of the Crohn’s Disease Elimination Diet (CDED) mentioned above did use the ModulenTM formula and it was a positive study.(19) As such, this might be considered to be the most evidence-based choice among the available formulas.
The formulas for consideration include EnsureTM, JevityTM, ResourceTM and ModulenTM, all of which are polymeric feeds that can be ingested orally. ModulenTM IBD is purported to have specific anti-inflammatory effects related to a high content of TGF-beta. As noted, it was the formula used in the Levine trial of CDED and PEN. VivonexTM is an elemental feed that would be difficult to ingest by mouth.
References
-
Grant A, Crane M, Laupacis A, et al. Engaging Patients and Caregivers in Research for Pediatric Inflammatory Bowel Disease: Top 10 Research Priorities. J Pediatr Gastroenterol Nutr. 2019;69(3):317–23.
-
Piovani D, Danese S, Peyrin-Biroulet L, et al. Environmental Risk Factors for Inflammatory Bowel Diseases: An Umbrella Review of Meta-analyses. Gastroenterology. 2019;157(3):647–59.
-
Khalili H, Håkansson N, Chan SS, et al. Adherence to a Mediterranean diet is associated with a lower risk of later-onset Crohn’s disease: results from two large prospective cohort studies. Gut. 2020; Jan 3 [Epub ahead of print].
-
Limketkai BN, Iheozor-Ejiofor Z, Gjuladin-Hellon T, et al. Dietary interventions for induction and maintenance of remission in inflammatory bowel disease. Cochrane Database Syst Rev. 2019;2:CD012839.
-
Ruemmele FM, Veres G, Kolho KL, et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J Crohn’s Colitis. 2014;8(10):1179–207.
-
Swaminath A, Feathers A, Ananthakrishnan AN, et al. Systematic review with meta-analysis: enteral nutrition therapy for the induction of remission in paediatric Crohn’s disease. Aliment Pharmacol Ther. 2017;46(7):645–56.
-
Pigneur B, Lepage P, Mondot S, et al. Mucosal Healing and Bacterial Composition in Response to Enteral Nutrition Vs Steroid-based Induction Therapy-A Randomised Prospective Clinical Trial in Children With Crohn’s Disease. J Crohns Colitis. 2019;13(7):846–55.
-
Lamb CA, Kennedy NA, Raine T, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68(Suppl 3):s1–s106.
-
Greenberg GR, Fleming CR, Jeejeebhoy KN, et al. Controlled trial of bowel rest and nutritional support in the management of Crohn’s disease. Gut. 1988;29(10):1309–15.
-
Heerasing N, Thompson B, Hendy P, et al. Exclusive enteral nutrition provides an effective bridge to safer interval elective surgery for adults with Crohn’s disease. Aliment Pharmacol Ther. 2017;45(5):660–9.
-
Pfeffer Gik T, Yanai H, Godny L, et al. Exclusive enteral nutrition in adults with active Crohn’s disease is associated with decreased disease activity [UEGW 2018 abstract P1875]. United European Gastroenterol J. 2018;6(8S):A746.
-
Lawley M, Wu JW, Navas-López VM, et al. Global Variation in Use of Enteral Nutrition for Pediatric Crohn Disease. J Pediatr Gastroenterol Nutr. 2018;67(2):e22–e29.
-
Sabino J, Lewis JD, Colombel JF. Treating Inflammatory Bowel Disease With Diet: A Taste Test. Gastroenterology. 2019;157(2):295–7.
-
Johnson T, Macdonald S, Hill SM, et al. Treatment of active Crohn’s disease in children using partial enteral nutrition with liquid formula: a randomised controlled trial. Gut. 2006;55(3):356–61.
-
Levine A, Sigall Boneh R, Wine E. Evolving role of diet in the pathogenesis and treatment of inflammatory bowel diseases. Gut. 2018;67(9):1726–38.
-
Cohen SA, Gold BD, Oliva S, et al. Clinical and mucosal improvement with specific carbohydrate diet in pediatric Crohn disease. J Pediatr Gastroenterol Nutr. 2014;59(4):516–21.
-
Suskind DL, Cohen SA, Brittnacher MJ, et al. Clinical and Fecal Microbial Changes With Diet Therapy in Active Inflammatory Bowel Disease. J Clin Gastroenterol. 2018;52(2):155–63.
-
Svolos V, Hansen R, Nichols B, et al. Treatment of Active Crohn’s Disease With an Ordinary Food-based Diet That Replicates Exclusive Enteral Nutrition. Gastroenterology. 2019;156(5):1354–67.
-
Levine A, Wine E, Assa A, et al. Crohn’s Disease Exclusion Diet Plus Partial Enteral Nutrition Induces Sustained Remission in a Randomized Controlled Trial. Gastroenterology. 2019;157(2):440–50.
-
Sigall Boneh R, Sarbagili Shabat C, Yanai H, et al. Dietary Therapy With the Crohn’s Disease Exclusion Diet is a Successful Strategy for Induction of Remission in Children and Adults Failing Biological Therapy. J Crohns Colitis. 2017;11(10):1205–12.
-
Clinicaltrials.gov. Diet for Induction and Maintenance of Remission and Re-biosis in Crohn’s Disease (DIETOMICS-CD). ClinicalTrials.gov Identifier: NCT02843100.
-
Personal correspondence, Dr. Arie Levine.
-
Mack DR, Benchimol EI, Critch J, et al. Canadian Association of Gastroenterology Clinical Practice Guideline for the Medical Management of Pediatric Luminal Crohn’s Disease. Gastroenterology. 2019;157(2):320–48.
-
Wall CL, Day AS, Gearry RB. Use of exclusive enteral nutrition in adults with Crohn’s disease: A review. World J Gastroenterol. 2013;19(43):7652–60.
-
Panaccione R, Steinhart AH, Bressler B. Canadian Association of Gastroenterology Clinical Practice Guideline for the Management of Luminal Crohn’s Disease. J Can Assoc Gastroenterol. 2019;2(3):e1–34.
-
Sigall-Boneh R, Levine A, Lomer M, et al. Research Gaps in Diet and Nutrition in Inflammatory Bowel Disease. A Topical Review by D-ECCO Working Group [Dietitians of ECCO]. J Crohns Colitis. 2017;11:1407–19.
-
Harries AD, Jones LA, Danis V, et al. Controlled trial of supplemented oral nutrition in Crohn’s disease. Lancet. 1983;1:887–90.
-
Hanai H, Iida T, Takeuchi K, et al. Nutritional therapy versus 6-mercaptopurine as maintenance therapy in patients with Crohn’s disease. Dig Liver Dis. 2012;44:649–54.
-
Takagi S, Utsunomiya K, Kuriyama S, et al. Effectiveness of a ‘half elemental diet’ as maintenance therapy for Crohn’s disease: A randomized controlled trial. Aliment Pharmacol Ther. 2006;24:1333–40.
-
Yamamoto T, Nakahigashi M, Saniabadi AR, et al. Impacts of long-term enteral nutrition on clinical and endoscopic disease activities and mucosal cytokines during remission in patients with Crohn’s disease: a prospective study. Inflamm Bowel Dis. 2007;13:1493–501.
-
Yamamoto T, Nakahigashi M, Umegae S, et al. Impact of long-term enteral nutrition on clinical and endoscopic recurrence after resection for Crohn’s disease: A prospective, non-randomized, parallel, controlled study. Aliment Pharmacol Ther. 2007;25:67–72.
-
Verma S, Kirkwood B, Brown S, Giaffer MH. Oral nutritional supplementation is effective in the maintenance of remission in Crohn’s disease. Dig Liver Dis. 2000;32:769–74.
-
Wilschanski M, Sherman P, Pencharz P, et al. Supplementary enteral nutrition maintains remission in paediatric Crohn’s disease. Gut. 1996;38:543–8.
-
Ghishan FK, Kiela PR. Vitamins and minerals in IBD. Gastroenterol Clin North Am. 2017;46(4):797–808.
-
Crohn’s and Colitis Foundation. Vitamin and Mineral Supplementation. Available on-line at: https://www.crohnscolitisfoundation.org/diet-and-nutrition/supplementation. Accessed November 2019.
-
Bernstein CN, Eliakim A, Fedail S, et al. World Gastroenterology Organisation Global Guidelines: Inflammatory Bowel Disease. Available on-line at: https://www.worldgastroenterology.org/guidelines/global-guidelines/inflammatory-bowel-disease-ibd/inflammatory-bowel-disease-ibd-english. Accessed November 2019.
-
Turner D, Shah PS, Steinhart AH, et al. Maintenance of remission in inflammatory bowel disease using omega-3 fatty acids (fish oil): a systematic review and meta-analyses. Inflamm Bowel Dis. 2011;17(1):336–45.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC AGAF, Director, Division of Gastroenterology, Professor, Department of Medicine, McMaster University, Hamilton, ON
Contributing Editor
Eytan Wine, MD PhD FRCPC, Associate Professor of Pediatrics and Physiology, Division of Pediatric Gastroenterology, University of Alberta, Edmonton, AB
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Anne M. Griffiths, MC FRCPC, University of Toronto, Toronto, ON
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
IBD Dialogue 2020·Volume 16 is made possible by unrestricted educational grants from…

![]()


Published by Catrile & Associates Ltd., 1B-391 Berkeley Street, Toronto, ON M5A 2X8
(c) Catrile & Associates Ltd., 2020. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.
