Rheumatological Manifestations in IBD
Rheumatological Manifestations in IBD
May 12, 2020
Issue 04
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by the contributing editor, Dr Glen Hazlewood, at the 20th annual national meeting, Mentoring in IBD XX: The Master Class, held November 15, 2019 in Toronto, Ontario.
Introduction
Up to 20% of patients with inflammatory bowel disease (IBD) have associated spondylarthritis (SpA).(1) Distinguishing SpA from non-inflammatory musculoskeletal pain and other immune-mediated inflammatory disorders requires a careful history, physical exam, appropriate laboratory investigations and imaging.(1)
The presence of SpA also introduces new therapeutic considerations and challenges in the treatment of IBD.
This summary reviews the clinical presentation, diagnosis and treatment of common rheumatologic conditions in patients with IBD, focusing on practical aspects for the gastroenterologist, and highlighting the importance of shared care between rheumatology and gastroenterology.
Distinguishing Between Causes of Joint Pain in IBD
The key question to be answered when faced with an IBD patient with joint pain is whether or not there is an inflammatory arthritis. In the peripheral joints, the key to making this distinction is determining whether or not there is joint swelling (indicative of synovitis). In the axial skeleton, for patients who present with back pain, swelling is not assessable, so other tools must be used.
Peripheral SpA
The classification criteria for peripheral SpA are quite straightforward. The patient must have one or more of arthritis, enthesitis or dactylitis, plus one or more of a number of comorbidities or indicators, of which IBD is one.(2) So for patients with IBD, it is simply a question of determining whether or not the patient has arthritis, enthesitis or dactylitis.
A key feature to make a diagnosis of inflammatory arthritis is the presence of joint swelling on physical exam. While a detailed musculoskeletal exam is typically beyond the scope of most gastroenterologists, there are some simple clues on history and exam that are helpful. On a practical level, patients can be asked if the joint looks swollen; many patients will report that a joint feels swollen even when it is not. Comparing the affected side to the unaffected side can also be informative. The patient should also be asked to demonstrate the range of motion for the joint(s) in question. If a full range of motion is possible, joint swelling is very unlikely. Also, if a patient can move a joint throughout the entire range of motion without pain, this is a very good sign that the pain is referred from elsewhere (e.g., pain in the shoulder may be referred from the neck). Using various types of range of motion assessments can also be helpful to determine the source of pain. If there is pain on active range of motion, but no pain on passive motion of the joint (i.e., the patient relaxes and you move the joint), then the problem is originating outside of the joint, typically from a tendon or other periarticular structure.
The time course of the symptoms may also be helpful. If the onset is quite defined and focal, this suggests an inflammatory arthritis, while insidious and widespread onset is more suggestive of other processes such as fibromyalgia. The status of IBD should be considered with the joint assessment, as activity in the peripheral joints tends to parallel activity in the gut. Morning stiffness can also be informative. People with inflammatory arthritis tend to have more than 30 minutes of morning stiffness. A helpful question to assess the duration of morning stiffness would be something like “How long does it take from when you wake up until you are as good as you are going to be that day?”
Enthesitis (inflammation at the site of tendon insertion into the bone) is also a common feature in patients with spondylarthritis and can be experienced in many sites throughout the body, but are most common in the heel (i.e., Achilles tendon). With respect to imaging, plain X-rays are not sensitive for enthesitis. MRI is very useful, but access is difficult. Ultrasound is also a useful tool, but identification of enthesitis does require an experienced technician.
Dactylitis refers to a swollen finger or toe, which is actually a combination of enthesitis and arthritis. The result is a digit that resembles a cocktail sausage. Even if not currently present, asking the patient if he or she has ever experienced this is a helpful part of the history.
Axial SpA
To make a diagnosis of axial SpA, the patient must have a history of three months or longer of back pain (with or without peripheral manifestations) and an age of onset younger than 45 years. In addition, there must either be evidence of sacroiliitis on imaging plus at least one other SpA feature (IBD is one of these). Alternatively, rather than evidence of sacroiliitis, the patient can have a positive test for HLA-B27 plus at least two of the SpA features.
Importantly, criteria for the diagnosis of inflammatory back pain,(3) are not the same as criteria for axial SpA, which is potentially confusing and should be kept in mind. The criteria for inflammatory back pain are all historical; the patient needs to have four or more of: age of onset 40 years or younger, insidious onset, improvement with exercise, no improvement with rest and pain at night (with improvement throughout the day). Within the group of individuals who meet these criteria, approximately 50–60% will have axial SpA.
Another potential red flag for axial SpA in the history is a report of alternating buttock pain.(4) Patients may also report this as hip pain. It is therefore important if a patient reports hip, back or buttock pain to ask them to demonstrate where exactly they are feeling the pain. Figure 1 shows the association of pain location with various diagnoses. Sacroiliac pain is felt posteriorly at the top of the buttock.
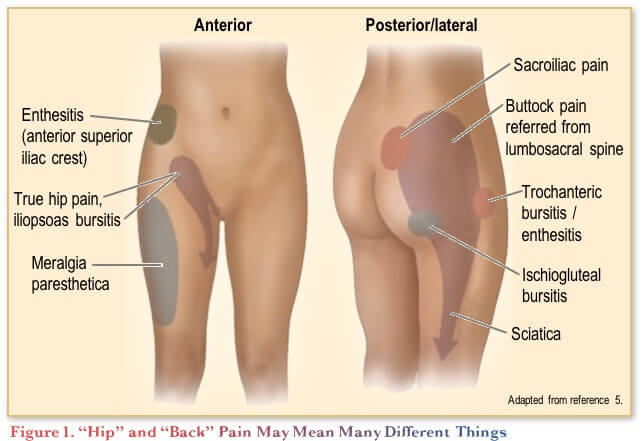
With this information in mind, the physical examination should include pressing on the area(s) the patient identifies as the source of the pain, to further help identify likely non-inflammatory causes. Other special manoeuvres that are used to assess spinal mobility (e.g., Schober’s test) are more helpful in following patients over time and have little role in diagnosis, owing to their poor sensitivity and specificity.
For imaging, MRI is the gold standard to identify inflammation in the spine and sacroiliac joints. Plain radiographs are typically done, but they are typically normal, unless a patient is presenting late.
Testing for HLA-B27 is quite helpful to raise suspicion of inflammatory disease. Approximately 50–70% of IBD patients with axial SpA are HLA-B27 positive, whereas only about 10% of IBD patients without SpA are HLA-B27 positive.(6)
Differential diagnoses
Fibromyalgia is a potential alternative cause of pain among people with IBD. The diagnostic criteria for this syndrome are: multi-site pain, moderate to severe sleep problems (or fatigue) and symptoms that have been present for at least three months.(7)
Widespread pain (both inside and outside the joints) is the hallmark of this condition and is the most useful feature to distinguish this from inflammatory arthropathies. Fatigue in general (even after a good night’s sleep) is another core characteristic of fibromyalgia, as is post-exertional fatigue. For the latter, asking patients what happens when they “overdo it” can be helpful.
TNF-antagonist-induced lupus is possible, and should be kept in mind for patients with articular manifestations, but also keep in mind that it is quite rare (about 1 in 1000 prevalence). The typical presentation is a rash and joint symptoms. The presence of a peripheral arthritis in a patient whose IBD is well controlled does raise the suspicion of TNF-antagonist-induced lupus. Here again, confirming that the joint pain is inflammatory is critical, as the majority of patients with joint pain when their bowel disease is well controlled will have fibromyalgia or a mechanical cause. A number of laboratory tests (e.g., ANA, ENA, dsDNA) are typically ordered when TNF-induced lupus is suspected. The TNF antagonist does not have to be stopped right away in patients with non-serious manifestations (e.g., joint pain, rash) in whom TNF-induced systemic lupus erythematosus (SLE) is suspected, as it is important to first confirm the diagnosis. Dermatology and/or rheumatology involvement is recommended when this is suspected.
Rheumatoid arthritis (RA) is also possible, but would be much less likely in a patient with known-IBD. In this setting, it can be difficult to distinguish RA from peripheral SpA, as they can look similar clinically. RA may be suspected if radiographs show joint erosions, or if the patient is rheumatoid factor or anti-CCP antibody positive, particularly at high titres.
Treatment Considerations for Patients with IBD and SpA
For axial SpA, exercise is a core component of overall management, and can be expected to provide slight improvements in function, disease activity and, possibly, pain control.(8,9)
Adjunctive treatments include topical nonsteroidal anti-inflammatory drugs (NSAIDs) for enthesitis, or for axial disease. Importantly for patients with concomitant IBD is the potential for NSAIDs to exacerbate the IBD. A meta-analysis of the use of NSAIDs among patients with IBD, which included mostly observational studies, did not find a consistent association between NSAIDs and risk of Crohn’s disease (CD) and ulcerative colitis (UC) exacerbation.(10) Two randomized, controlled trials (RCTs) with COX-2-inhibiting NSAIDs (celecoxib or etoricoxib) among patients with IBD similarly did not find an association between these treatments and exacerbation of IBD.(11,12) Based on the higher-level of evidence from RCTs and conflicting reports for non-selective NSAIDS, COX-2 inhibitors would typically be preferred as a first-line NSAID for patients with IBD.
Local glucocorticoid injections can be helpful for peripheral arthritis and sacroiliitis. Some entheses can also be injected, although injections of the Achilles, patellar and quadriceps tendons should be avoided due to the risk of tendon rupture.
Systemic glucocorticoids may also be used for peripheral disease, typically as a bridging strategy until other systemic medications become effective. A single intra-muscular injection of triamcinolone acetonide (e.g., 80–100 mg), for example, may provide relief for six weeks. For axial disease, there is limited evidence of efficacy, although a single RCT did show modest benefit in axial SpA with high-dose (50 mg/day) oral prednisolone.(13)
Conventional disease-modifying antirheumatic drugs (DMARDs) have a limited role in SpA, with no evidence of significant benefit in axial disease and only limited evidence of efficacy of methotrexate or sulfasalazine in peripheral SpA.(9,14)
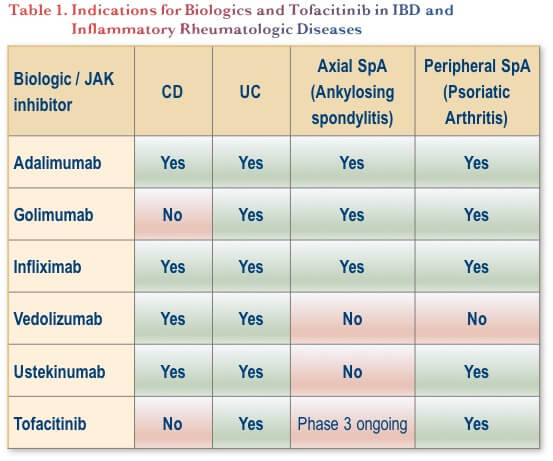
In terms of biologic therapy, anti-TNF agents (adalimumab, infliximab and golimumab) are indicated for the treatment of axial SpA.(15) For peripheral SpA, these agents are also indicated, as are ustekinumab, and tofacitinib (Table 1). Tofacitinib is also undergoing phase 3 study in axial disease.(16)
If a patient who is already on an anti-TNF therapy exhibits signs of active SpA, the first step would be to confirm that the SpA is indeed active, (by MRI for axial disease and by clinical diagnosis for peripheral disease). If the IBD is still controlled, preservation of the biologic is preferred if possible, so rheumatologists would typically try adjunctive or complementary measures first before considering switching therapy. This might include the measures listed above (e.g., local injections, NSAIDs, physiotherapy), and would depend on whether the patient has active axial disease, peripheral disease or both.
If the IBD is not controlled, NSAIDs should be avoided. Adding methotrexate or sulfasalazine might be considered, as would be switching to a different anti-TNF therapy or a biologic with another mechanism of action.
Combining biologics has been the focus of a number of clinical trials in rheumatoid arthritis, but these have not shown additional efficacy and have been associated with increased toxicities.(17) In IBD, there is emerging anecdotal experience with combining vedolizumab with other biologics (anti-TNF or ustekinumab) or with tofacitinib. These have been better tolerated, with the suggestion of additional efficacy.
Conclusions
When faced with an IBD patient with suspected SpA, history and physical examination are critical to help distinguish between inflammatory and non-inflammatory pain and to localize the pain. The joint examination does not have to be complicated; gastroenterologists can incorporate the simple recommendations from this review into their practices.
When inflammatory SpA is identified, there are several adjunctive or complementary approaches to try before one should consider switching a biologic. Collaboration with a rheumatologist is key to developing the best strategies to protect the bowels and the joints.
Clinical Case
This is a case of a 33-year-old woman with an eight-year history of CD. She has two young children (toddler and baby). She has noticed increasing morning back pain, which is causing poor quality of life, and a mild increase in gastrointestinal (GI) symptoms which she only admits to on extended questioning. GI symptoms include three semi-formed bowel movements (BMs)/day with no bleeding or abdominal pain. She has been on azathioprine 100 mg/day for the last five years and has had previous exposure to 5-ASA. She has had one prior course of oral corticosteroids prior to azathioprine maintenance therapy, no hospitalizations, no perianal disease, and no skin or eye manifestation.
Commentary
At this point, further investigation is warranted to evaluate both her back pain and her CD.
With respect to the history, one would want to know more details about the severity of her pain and its impact on her daily functioning. This is her primary complaint and although you are not a back specialist of any kind, she is in front of you asking about this and she may not see another physician for this problem in a timely manner.
The back pain could be due to a number of possibilities, but there is a known association between IBD and axial spondyloarthritis (SpA: e.g., ankylosing spondylitis). Most sources estimated that axial arthropathies occur in 3% to 5% of patients with IBD, although frequencies of up to 25% have been reported.(1)
Because IBD can also be associated with peripheral SpA, you could also ask if there has been any other pain in the peripheral joints. Peripheral arthralgias/arthritis affect an estimated 5% to 10% of patients with UC and 10% to 20% of those with CD.(1) With respect to the physical examination, in addition to your typical examination, you could look for joint swelling and observe her range of motion and pain from routine movements. Examining the fingers for evidence of dactylitis is also important.
Her increasing GI symptoms also warrant further workup at this point. Routine laboratory assessments are appropriate here (e.g., hemoglobin, MCV, CRP, fecal calprotectin, stool cultures). A colonoscopy is also warranted to investigate disease activity.
Case Evolution
On examination, her vital signs are stable, abdominal examination is unremarkable, and you wonder whether she is exhibiting back stiffness (but as a gastroenterologist, you are not quite sure how to examine a back). No perianal disease. Other joints don’t demonstrate obvious ‘swelling’, and there are no rashes. You forget to examine her eyes.
A colonoscopy is performed. This demonstrates some patchy erythema in the descending colon, ascending colon and cecum, with small erosions particularly on the right side, and aphthae in the terminal ileum for at least 10 cm without any evidence of stenosis.
Laboratory tests:
- Hb: 108 g/L
- MCV: 81 fL
- CRP: 2.0 mg/L
- Fecal calprotectin: 98 mcg/g
- Stool cultures: Negative for C. difficile toxin, C&S, O&P.
The patient is not concerned regarding her GI symptoms but wants you or another doctor to fix her back pain, as she is having trouble picking up and carrying the kids.
Commentary
If this patient has access to a rheumatologist, referral for specialist work-up of her back pain would be a good idea at this point.
A recent joint consensus paper by a group of Italian gastroenterologists and rheumatologists identified several major “red flags” that should warrant referral of an IBD patient to a rheumatologist:(18)
- Chronic low back pain
- Dactylitis
- Enthesitis
- Pain/swelling of peripheral joints
A similar paper by a Spanish group identified the following major criteria and recommended referral for screening for SpA if any of these were present in a person with IBD:(19)
- Onset of chronic low back pain before 45 years of age
- Inflammatory low back pain or alternating buttock pain
- HLA-B27 positivity
- Sacroiliitis on imaging
- Arthritis
- Heel enthesitis
- Dactylitis
Case Evolution
You are increasingly concerned regarding her CD (you are a gastroenterologist after all)! You had already arranged her pre-biologic work up when she first attended for consultation three years ago, and she is up to date with her vaccinations. You wish to talk to her about the choice of biologics while the above ‘joint’ work up is pending.
Commentary
For her CD, she is currently uncontrolled despite maintenance azathioprine. Switching to or adding a biologic is a guideline-recommended option at this point.(20)
Given the context of the current chronic back pain, although you do not yet have a diagnosis of inflammatory arthritis, you nonetheless consider this a distinct possibility.
Notably, consensus guidelines for the treatment of extra-intestinal manifestations of IBD state that “sulfasalazine and methotrexate are of limited efficacy [for axial SpA].”(21)
You mention your concern about potential comorbid axial SpA to your patient and tell her that there are potent therapies available that can treat her CD and can also improve her joint symptoms if they are indeed inflammatory in nature (e.g., axial SpA). In Canada, the only two biologics that are indicated for both CD and ankylosing spondylitis are the anti-TNF agents adalimumab and infliximab. Whether or not to continue with the azathioprine when initiating a biologic remains a matter of debate. Either biologic monotherapy or combination therapy is endorsed by guidelines.(20)
Case Evolution
You decide to start a biologic. Much is discussed regarding the choice of a biologic. On further questioning, your patient states her mother has multiple sclerosis and her father is being treated for lymphoma. Further, she would prefer a treatment that is provided by subcutaneous injection rather than infusion.
Commentary
With respect to the decision of whether or not to continue the azathioprine, although guidelines leave this to the discretion of the patient and clinician, the family history of lymphoma may tip the scales in favor of moving to anti-TNF monotherapy. There is a known increased risk of lymphoma among IBD patients treated with thiopurines.(22,23)
With respect to the choice of individual biologic, each of the listed agents has demonstrated clinical trial efficacy for CD, with no head-to-head studies having yet been completed.(24–27)
As mentioned above, perhaps a key motivator for choosing a biologic at this point would be the possibility of an inflammatory rheumatologic comorbidity. Both adalimumab and infliximab have demonstrated efficacy in RCTs in ankylosing spondylitis and are approved in Canada to treat that condition.(28,29)
The IL-12/23 antagonist ustekinumab showed promise in a phase 2 study in ankylosing spondylitis,(30) but in phase 3 study did not meet primary endpoints and the clinical research program was halted for this indication.(31)
The gut-specific anti-integrin vedolizumab has not been studied in SpA. Use of this agent may lead to improvements in extraintestinal manifestations, but there is also the risk of paradoxical worsening of joints and/or skin.(32)
In small case series, the combination of an anti-TNF and vedolizumab has been efficacious and safe for patients with comorbid IBD and SpA.
The patient indicated she would prefer subcutaneous administration. Adalimumab seems to be the best choice for her among the available biologics.
Case Evolution
The patient finally agrees on and chooses adalimumab, but after much resistance. She chose to stop the azathioprine on her own accord (without your knowledge), as she felt it was worsening her back pain. On adalimumab monotherapy, her gastrointestinal symptoms much improved and by week 16, her fecal calprotectin has dropped to 243 mcg/g. Her back pain is still present, but improved; however, she now has a ‘rash’ on her face and arms, says her hands are ‘swollen’ and is insisting on stopping all therapy, including the adalimumab.
Commentary
You consider the possibility that the skin and joint symptoms could be drug-induced lupus. Laboratory tests to consider here would be ANA, ENA, dsDNA.(33) You are also still awaiting the results of the MRI.
Case Evolution
You remember to review the MRI of the spine, and it does show evidence of sacroiliitis. Her ANA is positive but dsDNA and anti-histone antibodies are negative. The dermatologist and rheumatologist are not convinced that the nature of her rash or joint symptoms are consistent with drug-induced lupus, but the patient is insistent on stopping all therapy and seeing a new gastroenterologist, rheumatologist and dermatologist. She is not interested in biologics, let alone, combination biologic therapy.
References
-
Vavricka SR, Schoepfer A, Scharl M, et al. Extraintestinal Manifestations of Inflammatory Bowel Disease. Inflamm Bowel Dis. 2015;21(8):1982–92.
-
Rudwaleit M, van der Heijde D, Landewé R, et al. The Assessment of SpondyloArthritis International Society classification criteria for peripheral spondyloarthritis and for spondyloarthritis in general. Ann Rheum Dis. 2011;70(1):25–31.
-
Sieper J, van der Heijde D, Landewé R, et al. New criteria for inflammatory back pain in patients with chronic back pain: a real patient exercise by experts from the Assessment of SpondyloArthritis international Society (ASAS). Ann Rheum Dis. 2009;68(6):784–8.
-
Vanelderen P, Szadek K, Cohen SP, et al. 13. Sacroiliac joint pain. Pain Pract. 2010; 10(5):470–8.
-
Cush JJ. Chapter 363: Approach to Articular and Musculoskeletal Disorders. In: Jameson J, et al (eds). Harrison’s Principles of Internal Medicine, 20e. 2019. Online at https://accessmedicine.mhmedical.com/content.aspx?bookid=2129§ionid=192285968, accessed April 9, 2020.
-
Ossum AM, Palm Ø, Lunder AK, et al. Ankylosing Spondylitis and Axial Spondyloarthritis in Patients With Long-term Inflammatory Bowel Disease: Results From 20 Years of Follow-up in the IBSEN Study. J Crohns Colitis. 2018;12(1):96–104.
-
Arnold LM, Bennett RM, Crofford LJ, et al. AAPT Diagnostic Criteria for Fibromyalgia. J Pain. 2019;20(6):611–28.
-
Regnaux JP, Davergne T, Palazzo C, et al. Exercise programmes for ankylosing spondylitis. Cochrane Database Syst Rev. 2019;10:CD011321.
-
Ward MM, Deodhar A, Gensler LS, et al. 2019 Update of the American College of Rheumatology/Spondylitis Association of America/Spondyloarthritis Research and Treatment Network Recommendations for the Treatment of Ankylosing Spondylitis and Nonradiographic Axial Spondyloarthritis. Arthritis Rheumatol. 2019;71(10):1599–613.
-
Moninuola OO, Milligan W, Lochhead P, et al. Systematic review with meta-analysis: association between acetaminophen and nonsteroidal anti-inflammatory drugs (NSAIDs) and risk of Crohn’s disease and ulcerative colitis exacerbation. Aliment Pharmacol Ther. 2018;47(11):1428–39.
-
Sandborn WJ, Stenson WF, Brynskov J, et al. Safety of celecoxib in patients with ulcerative colitis in remission: a randomized, placebo-controlled, pilot study. Clin Gastroenterol Hepatol. 2006;4(2):203–11.
-
El Miedany Y, Youssef S, Ahmed I, et al. The gastrointestinal safety and effect on disease activity of etoricoxib, a selective cox-2 inhibitor in inflammatory bowel diseases. Am J Gastroenterol. 2006;101(2):311–7.
-
Haibel H, Fendler C, Listing J, et al. Efficacy of oral prednisolone in active ankylosing spondylitis: results of a double-blind, randomised, placebo-controlled short-term trial. Ann Rheum Dis. 2014;73(1):243–6.
-
Singh JA, Guyatt G, Ogdie A, et al. Special Article: 2018 American College of Rheumatology/National Psoriasis Foundation Guideline for the Treatment of Psoriatic Arthritis. Arthritis Rheumatol. 2019;71(1):5–32
-
Health Canada. Drug product database. Online at https://health-products.canada.ca/dpd-bdpp/index-eng.jsp. Accessed April 9, 2020.
-
gov. Efficacy and Safety of Tofacitinib in Subjects With Active Ankylosing Spondylitis (AS). ClinicalTrials.gov Identifier: NCT03502616.
-
Hirten RP, Iacucci M, Shah S, et al. Combining Biologics in Inflammatory Bowel Disease and Other Immune Mediated Inflammatory Disorders. Clin Gastroenterol Hepatol. 2018;16(9):1374–84.
-
Felice C, Leccese P, Scudeller L, et al. Red flags for appropriate referral to the gastroenterologist and the rheumatologist of patients with inflammatory bowel disease and spondyloarthritis. Clin Exp Immunol. 2019;196(1):123–38.
-
Sanz Sanz J, Juanola Roura X, Seoane-Mato D, et al. Screening of Inflammatory Bowel Disease and Spondyloarthritis for Referring Patients Between Rheumatology and Gastroenterology. Gastroenterol Hepatol. 2018;41(1):54–62.
-
Panaccione R, Steinhart AH, Bressler B. Canadian Association of Gastroenterology Clinical Practice Guideline for the Management of Luminal Crohn’s Disease. J Can Assoc Gastroenterol. 2019;2(3):e1–34.
-
Harbord M, Annese V, Vavricka SR, et al. The First European Evidence-based Consensus on Extra-intestinal Manifestations in Inflammatory Bowel Disease. J Crohn’s Colitis. 2016;10(3):239–54.
-
Kotlyar DS, Lewis JD, Beaugerie L, et al. Risk of lymphoma in patients with inflammatory bowel disease treated with azathioprine and 6-mercaptopurine: a meta-analysis. Clin Gastroenterol Hepatol. 2015;13(5):847–58.
-
Osterman MT, Sandborn WJ, Colombel JF, et al. Increased risk of malignancy with adalimumab combination therapy, compared with monotherapy, for Crohn’s disease. Gastroenterology. 2014; 146(4):941–9.
-
Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn’s disease: the CHARM trial. Gastroenterology. 2007;132(1):52–65.
-
Colombel JF, Sandborn WJ, Reinisch W, et al. Infliximab, azathioprine, or combination therapy for Crohn’s disease. N Engl J Med. 2010;362(15):1383–95.
-
Sandborn WJ, Feagan BG, Rutgeerts P, et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N Engl J Med. 2013; 369(8):711–21.
-
Feagan BG, Sandborn WJ, Gasink C, et al. Ustekinumab as Induction and Maintenance Therapy for Crohn’s Disease. N Engl J Med. 2016;375(20):1946–60.
-
Revicki DA, Luo MP, Wordsworth P, et al. Adalimumab reduces pain, fatigue, and stiffness in patients with ankylosing spondylitis: results from the adalimumab trial evaluating long-term safety and efficacy for ankylosing spondylitis (ATLAS) J Rheumatol. 2008;35(7):1346–53.
-
van der Heijde D, Dijkmans B, Geusens P, et al. Efficacy and safety of infliximab in patients with ankylosing spondylitis: results of a randomized, placebo-controlled trial (ASSERT). Arthritis Rheum. 2005;52(2):582–91.
-
Poddubnyy D, Hermann K-GA, Callhoff J, et al. Ustekinumab for the treatment of patients with active ankylosing spondylitis: results of a 28-week, prospective, open-label, proof-of-concept study (TOPAS). Ann Rheum Dis. 2014;73:817–23.
-
Deodhar A, Gensler LS, Sieper J, et al. Three Multicenter, Randomized, Double-Blind, Placebo-Controlled Studies Evaluating the Efficacy and Safety of Ustekinumab in Axial Spondyloarthritis. Arthritis Rheumatol. 2019;71(2):258–70.
-
Tadbiri S, Peyrin-Biroulet L, Serrero M, et al. Impact of vedolizumab therapy on extra-intestinal manifestations in patients with inflammatory bowel disease: a multicentre cohort study nested in the OBSERV-IBD cohort. Aliment Pharmacol Ther. 2018;47(4):485–93.
-
Dalle Vedove C, Simon JC, Girolomoni G. Drug-induced lupus erythematosus with emphasis on skin manifestations and the role of anti-TNFα agents. J Dtsch Dermatol Ges. 2012;10(12):889–97.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC AGAF, Director, Division of Gastroenterology, Professor, Department of Medicine, McMaster University, Hamilton, ON
Contributing Editor
Glen S. Hazlewood, MD PhD FRCPC, Assistant Professor, Departments of Medicine and Community Health Sciences, Cumming School of Medicine, University of Calgary, Calgary, AB
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Anne M. Griffiths, MC FRCPC, University of Toronto, Toronto, ON
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
IBD Dialogue 2020·Volume 16 is made possible by unrestricted educational grants from…
![]()



Published by Catrile & Associates Ltd., 1B-391 Berkeley Street, Toronto, ON M5A 2X8
(c) Catrile & Associates Ltd., 2020. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.
