Approaches to Dysplasia
Approaches to Dysplasia
March 1, 2021
Issue 02
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by Dr. Talat Bessissow, at the 22nd annual national meeting, Mentoring in IBD XXII: The Master Class, held virtually, November 5, 2021.
Introduction: Colon Cancer Risk
This presentation provided a brief summary of the overall approach to dysplasia surveillance in inflammatory bowel disease (IBD), reviewed current and novel techniques, and then discussed the endoscopic management of dysplasia.
Current dysplasia surveillance practices stem from the evidence demonstrating the increased risk of colorectal cancer (CRC) in patients with IBD. A seminal meta-analysis by Eaden et al demonstrated a high risk of CRC in long-standing ulcerative colitis (UC; after 10 years of disease).(1) A more recent database study with 178 million person-years of follow-up showed that the relative risk for CRC was not increased in UC patients in general, however subgroup analyses identified that patients with UC and primary sclerosing cholangitis (PSC) and those with long-standing disease (>8 years) were specifically at higher risk.(2)
To investigate this further, a study looking at cumulative incidence across 10-year intervals of disease duration found that the incidence risk increased to 6.7% after 30 years and 13.6% after 50 years.(3) The same study looked at incidence rates per decade from 1973 to 2012, and found a decrease in incidence over time but an increase from 2003–12.(3) Hypotheses for this increase include improved adherence to surveillance and increased sensitivity of detection methods. This aligns with a shift toward finding less advanced cancer, suggesting earlier detection of CRC. Their analysis found that the probability of remaining CRC-free is similar after detection of low-grade dysplasia (LGD) and indefinite dysplasia, but higher as expected after detection of high-grade dysplasia (HGD).(3)
Studies have also established that risk factors for dysplasia include extensive colitis, long-standing inflammation, colonic stricture, PSC, prior dysplasia, and family history of CRC (particularly aged <50 years). In contrast, protective factors for dysplasia include surveillance colonoscopy, 5-ASA, thiopurines, and smoking(4). It was noted that any medication that can provide endoscopic or histologic healing likely decreases risk, independent of a specific chemopreventive effect.
Surveillance Techniques
Surveillance techniques have changed significantly over time from standard white light endoscopy (WLE), to zoom and high-definition endoscopy. Additional modalities include dye-spray chromoendoscopy (DCE), electronic virtual chromoendoscopy (VCE) and confocal laser endomicroscopy, although the latter is not widely used.
Regardless of the technique, important fundamentals for dysplasia detection remain consistent including conducting surveillance in endoscopic remission, use of a HD scope, washing and careful inspection of the mucosa, and good bowel preparation. Target biopsies are taken from suspicious mucosal abnormalities or site of prior dysplasia with endoscopic resection preferred if well demarcated.
With respect to biopsy protocols, data from Van den Broek and colleagues(5) showed a much higher yield with targeted biopsies versus a standard random biopsy protocol. Furthermore, challenges with adherence with time-consuming random biopsy protocols question their utility. To better understand the role of random biopsies, The IBD-Dysplasia Study is currently underway. This Canadian randomized, parallel-group, non-inferiority trial will compare random and targeted biopsies to targeted biopsies alone for neoplasia detection during screening colonoscopy in adult persons with colonic IBD.(6)
Dye-spray chromoendoscopy (DCE)
DCE efficacy has been evaluated in multiple studies. A meta-analysis of DCE in UC showed a 44% increase in detection of lesions, with a 27% increase in flat lesions compared to WLE.(7) These findings were reproduced in other meta-analyses.(8,9)
Virtual chromoendoscopy (VCE)
Digital modalities provide enhanced chromoendoscopy images without the use of dye. Data supporting their efficacy include a meta-analysis showing virtual chromoendoscopy as superior compared to standard WLE, but not compared to HD-WLE.(10) When they looked specifically at i-scan or narrow-band imaging (NBI) vs. dye spray chromoendoscopy, there was no difference between the modalities, suggesting it is a reasonable alternative.(10) Advantages of virtual chromoendoscopy include its ‘push the button’ application, ease of use in difficult colonoscopies, shorter withdrawal time (26.87 ± 9.89 minutes for CE vs. 15.74 ± 5.62 minutes for NBI, p<0.01), and lack of need for dye spraying or extra equipment.(11)
Dysplasia Management
An important American Gastroenterological Association (AGA) Clinical Practice Update on colorectal dysplasia in IBD was recently published and one of its key inclusions was guidance on timing of surveillance colonoscopies based on risk factors (see Table 1).(12)

While terminology to describe lesions continues to evolve, what is most clinically relevant is to determine if the lesion is resectable. Resectable lesions have good delineation of borders, no deep invasion, and adequate submucosal lift. It is recommended to use a common language when describing lesions based on the Paris classification in addition to describing the lesion’s size, morphology, border clarity, ulceration, location, activity of local colitis, completeness of resection, and any special techniques required for visualization. Lastly, it important to identify the Kudo pit pattern classification.(12)
With respect to post-resection recurrence, a metanalysis of recurrent CRC after endoscopic resection of polypoid lesions in IBD found a risk of 0.5% per year,(13) which is why these patients are considered high risk and continued surveillance colonoscopy is recommended annually for 5 years.(12)
Management of identified lesions is based on characterization, and an algorithm has been developed providing stepwise guidance (see Figure 1).
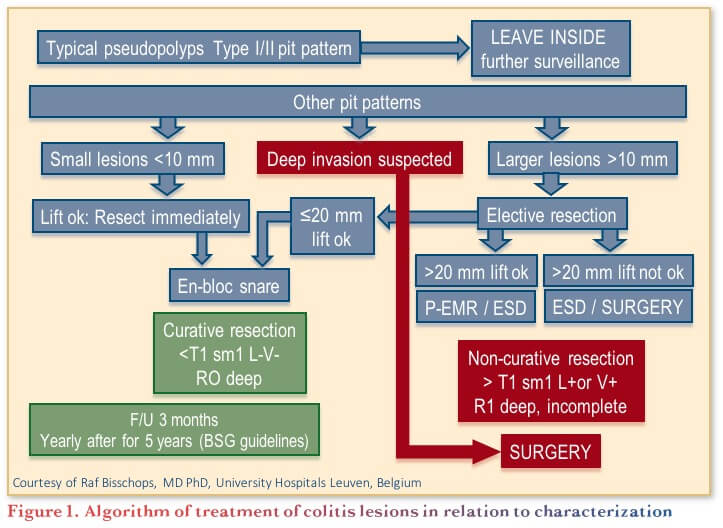
Conclusions
In conclusion, there is an increased risk of dysplasia in longstanding colonic inflammatory bowel disease, and this is highest in patients with concomitant PSC. Chromoendoscopy is the method of choice for surveillance, with VCE providing a good alternative. Practical guidance is available to help clinicians assess resectability and manage dysplasia based on lesion characteristics.
Clinical Case
Neil is a 25-year-old male with a known history of UC, extending to the hepatic flexure. He was diagnosed 5 years ago and was initially treated with prednisone and 5-ASA, but had a
flare within the first year so was started on vedolizumab. Currently, he is clinically in remission on a dose of 300 mg every 8 weeks with 1–2 formed stools daily, without blood.
Investigations:
- CBC: normal
- FCP: 162 mg/kg
- ALP is elevated
You perform a magnetic resonance cholangiopancreatography (MRCP), which confirms your suspicion of PSC. You explain to him that he is at increased risk of developing colon cancer and book him for a colonoscopy to screen for dysplasia.
Commentary
The 2021 AGA Clinical Practice Update recommends:
- Performing initial colonoscopy screening for dysplasia 8–10 years after disease diagnosis in all people with colonic IBD, and immediately on diagnosis of PSC.(12)
- DCE performed by appropriately trained endoscopists, is recommended particularly if a standard-definition endoscope is used or if there is a history of dysplasia, with VCE as a suitable alternative.(12)
Case Evolution
He was prepared for his colonoscopy with a split-dose PEG preparation. The preparation is quite good, but you wash a few small areas to improve visualization. You decide to use methylene blue for chromoendoscopy (as your endoscopy unit does not stock indigo carmine), 0.04–0.1% concentration in the waterjet.
Commentary
The 2021 AGA Clinical Practice Update recommends:
- Targeted biopsies be performed if mucosal findings are suspicious for dysplasia or are inexplicably different from the surrounding mucosa.(12)
- Endoscopic resection is preferred to biopsies when lesions are clearly demarcated without stigmata of invasive cancer or submucosal fibrosis.
- Describing precancerous colorectal lesions in IBD using a modified Paris classification.(12)
- Visible precancerous lesions should be further described based on size, morphology, clarity of borders, presence of ulceration, location, presence within an area of past or current colitis, perceived completeness of resection, and whether any special techniques were used for visualization.(12,14)
- The “five S” features have been proposed to describe colonic lesions (shape, size, site, surface [Kudo pit pattern], and surrounding mucosal activity) and other lesions.(14)
Case Evolution
As you are using chromoendoscopy, you take staging/mapping biopsies to assess for disease activity as well as targeted biopsies for suspicious areas. You use the Paris classification to describe any suspicious areas. You identify a 12 mm flat lesion in the transverse colon and resect. No other lesions were found.
Commentary
- The 2021 AGA Clinical Practice Update recommends that, considering the improved visibility with currently available HD endoscopes, unless concerns arise regarding a lesion’s borders and completeness of resection, routine biopsies of the flat mucosa surrounding a resected lesion are not needed, as they rarely demonstrate residual dysplasia.(12)
- Placement of a tattoo is recommended whenever a larger polyp is removed in order to aid in locating the lesion and future surveillance. Additional biopsies of the flat mucosa surrounding the site is also recommended to aid in the evaluation of adjacent dysplasia.(15)
Case Evolution
As you are confident that you resected the entire polyp, you do not take biopsies surrounding the polyp. Histology confirms HGD.
Commentary
The AGA 2021 Clinical Practice Update recommends:(12)
- Clearly delineated dysplastic-appearing lesions without stigmata of invasive cancer or significant submucosal fibrosis be considered for endoscopic resection.
- If there is uncertainty about the most appropriate management strategy, consider case discussion in multidisciplinary rounds or referral to a specialized IBD center.
- Follow-ups:(12)
- For resectable lesions <2 cm with no histological features of invasive cancer: Continued surveillance with colonoscopy planned for 3–12 months later depending on the size and grade of the dysplasia.
- For lesions ³2 cm, complex lesions, incomplete resection after several attempts or local recurrence: Intensive surveillance with colonoscopy planned every 3–6 months for the first year, with decision to resect based on lesion details, local expertise, and disease activity.
Case Evolution
You have a discussion with Neil and his wife and provide options of intensive surveillance every 6 months vs. colectomy. He elects to have intensive surveillance. At the next colonoscopy, you perform targeted and non-targeted biopsies and discover unifocal LGD on non-targeted biopsies.
Commentary
- The AGA 2021 Clinical Practice Update recommends surgery for lesions unresectable due to size, location, features of invasive cancer or submucosal fibrosis or invasive cancer on histology.(12)
- More recent data suggest that the risk of future CRC in the setting of unifocal low-grade dysplasia is considerably lower than in previous reports(16) and have declined substantially over time, likely due to better inflammatory control, improved endoscope resolution, improved bowel preparation, and better overall endoscopy quality.(3,17,18)
- Therefore, it is recommended to have a risk–benefit discussion with patients, taking disease control, and short- and long-term cancer risk factors into account.
Case Conclusion
You have a discussion with Neil. Given his brother also has PSC and colitis and is doing well with a colectomy and ileal pouch-anal anastomosis (IPAA), he elects to have surgery.
References
-
Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48(4):526–35.
-
Jess T, Simonsen J, Jorgensen KT, et al. Decreasing risk of colorectal cancer in patients with inflammatory bowel disease over 30 years. Gastroenterology. 2012;143(2):375–81.
-
Choi CHR, Rutter MD, Askari A, et al. Fourty-year analysis of colonoscopic surveillance program for neoplasia in ulcerative colitis: An updated overview. Am J Gastroenterol. 2015;110:1022–34.
-
Wijnands AM, de Jong ME, Lutgens MWMD, et al. Prognostic factors for advanced neoplasia in inflammatory bowel disease: A systematic review and meta-analysis. Gastroenterology. 2021;160(5):1584–98.
-
van den Broek F, Stokkers PC, Reitsma JB, et al. Random biopsies taken during colonoscopic surveillance of patients with longstanding ulcerative colitis: low yield and absence of clinical consequences. Am J Gastroenterol. 2014;109(5):715–22.
-
Canadian IBD Research Consortium. IBD-Dysplasia. Available at: https://circ-ccrm.ca/ibd-dysplasia/. Accessed: January 2022.
-
Subramanian V, Mannath J, Ragunath K, et al. Meta-analysis: the diagnostic yield of chromoendoscopy for detecting dysplasia in patients with colonic inflammatory bowel disease. Aliment Pharmacol Ther. 2011;33:304–12.
-
Laine L, Kaltenbach T, Barkun A, et al. SCENIC international consensus statement on surveillance and management of dysplasia in inflammatory bowel disease. Gastrointest Endosc. 2015;81(3):489–501.e26
-
Iannone A, Ruospo M, Wong G, et al. Chromoendoscopy for surveillance in ulcerative colitis and Crohn’s disease: A systematic review of randomized trials. Clin Gastroenterol Hepatol. 2017;15:1684–97.
-
El-Dallal M, Chen Y, Lin Q, et al. Meta-analysis of virtual-based chromoendoscopy compared with dye-spraying chromoendoscopy standard and high definition white light endoscopy in patients with inflammatory bowel disease at increased risk of colon cancer. Inflamm Bowel Dis. 2020;26:1319–29.
-
Bisschops R, Bessissow T, Joseph JA, et al. Chromoendoscopy versus narrow band imaging in UC: a prospective randomised controlled trial. Gut. 2018;67(6):1087–94.
-
Murthy SK, Feuerstein JD, Nguyen GC, et al. AGA Clinical Practice Update on Endoscopic Surveillance and Management of Colorectal Dysplasia in Inflammatory Bowel Diseases: Expert Review. 2021;161(3):1043–51.
-
Wanders LK, Dekker E, Pullens B, et al. Cancer risk after resection of polypoid dysplasia in patients with longstanding ulcerative colitis: a meta-analysis. Clin Gastroenterol Hepatol. 2014;12:756–64.
-
Adamina M, Feakins R, Iacucci M, et al. ECCO topical review optimising reporting in surgery, endoscopy, and histopathology. J Crohns Colitis. 2021;15(7):1089–105.
-
Clarke WT, Feuerstein JD. Colorectal cancer surveillance in inflammatory bowel disease: Practice guidelines and recent developments. World J Gastroenterol. 2019;25(30):4148–57.
-
Fumery M, Dulai PS, Gupta S, et al. Incidence, risk factors, and outcomes of colorectal cancer in patients with ulcerative colitis with low-grade dysplasia: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2017;15:665–74.
-
Wan J, Wang X, Yang ZP, et al. Systematic review with meta-analysis: chromoendoscopy versus white light endoscopy in detection of dysplasia in patients with inflammatory bowel disease. J Dig Dis. 2019;20:206–14.
-
Rex DK, Schoenfeld PS, Cohen J, et al. Quality indicators for colonoscopy. Gastrointest Endosc. 2015;81:31–53.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC AGAF, Director, Division of Gastroenterology, Professor, Department of Medicine, McMaster University, Hamilton, ON
Contributing Author
Talat Bessissow, MD CM FRCPC, Professor of Medicine, Division of Gastroenterology, McGill University Health Centre, Montreal, QC
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Anne M. Griffiths, MC FRCPC, University of Toronto, Toronto, ON
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
IBD Dialogue 2022·Volume 18 is made possible by unrestricted educational grants from…
![]()
![]()
![]()



![]()

Published by Catrile & Associates Ltd., 167 Floyd Avenue, East York, ON M4J 2H9
(c) Catrile & Associates Ltd., 2022. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.
