Thromboembolic Complications & IBD
Thromboembolic Complications & IBD
April 25, 2023
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by Dr. Mark Crowther, at the 23rd annual national meeting, Mentoring in IBD XXIII: The Master Class, held in Toronto on November 4, 2022.
Volume 19 | Issue 01
Introduction: IBD and the Risk of Thromboembolic Events
The objectives of this presentation were to review some of the more unusual presentations of venous thrombotic events (VTE) in IBD patients, discuss whether therapies for IBD mitigate this VTE risk, and to explore how to manage anticoagulation in patients with active IBD.
IBD is associated with a twofold greater risk of VTE. Specifically, a meta-analysis of 11 studies calculated a relative risk (RR) of 2.20 (95% CI 1.83–2.65) for deep vein thrombosis (DVT) and pulmonary embolism (PE) in those with IBD vs. without IBD.(1) Looking more specifically at IBD subtypes, they found a RR of 2.57 (95% CI 2.02–3.28) for ulcerative colitis (UC) and 2.12 (95% CI 1.40–3.20) for Crohn’s disease (CD).(1) Risk appears to correlate with multiple coincident risk factors including disease activity, systemic inflammation, immobilization, line insertion, medication administration, and the need for surgical or other interventions.(2-5) Lastly, patients often do not receive prophylactic anticoagulation therapy because of their IBD, which further increases their risk.
It is important to be aware that IBD also increases the risk of arterial thrombotic events (ATE), albeit this is not as strong an association as for VTE. Specifically, a population-based study from Copenhagen County that included 108,789 participants (of whom 1,203 had IBD) found a prevalence of cardiovascular disease of 13.2% in IBD patients vs.10.9% in the general population (p=0.009).(6) The authors suggest the increased risk may be due to the fact that IBD patients had higher levels of C-reactive protein (CRP) and fibrinogen, which may reflect chronic systemic inflammation as a risk factor.(6)
Mitigation Strategies for Patients with IBD
Given the risk of thromboembolic complications in IBD, a consensus paper that provides guidance on the prevention of A/VTE in patients with IBD was recently published.(3) In terms of mitigating risk in patients with IBD, it is recommended to identify risk factors and ones that can be ameliorated, including using prophylaxis when indicated, even in some ambulatory patients. Importantly, DVT prophylaxis should be provided with hospital admission, despite a patient having IBD, particularly for patients with acute disease, and there should be a very good reason documented if it is not provided.
In addition, it is important to remember that anything that helps control the IBD will decrease the risk of A/VTE when combined with pharmacological prophylaxis.(3) Specifically, 5-ASA derivatives may decrease the thromboembolic risk, while steroids may be associated with increased risk.(3,7-10) Other “standard of practice” mitigation strategies should be aggressively pursued such as mobilization. Meanwhile, there is no role for routine thrombophilia testing, as the results will not impact the choice of anticoagulation strategy.
Anticoagulation Options
There are a wide range of anti-coagulation options to consider.(11) Unfractionated heparin (UFH) still has an important role as, while it is complex to administer, it is the only reversible option and is not dependent on renal clearance, so is widely used with those with renal insufficiency. Low-molecular weight heparin (LMWH) has a predictable anticoagulant effect, is easy to administer, but is irreversible, and has some degree of renal dependence. LMWH is widely used in patients with renal failure and those with a high risk of bleeding despite its renal dependence and irreversible effects. Notably, warfarin still has a place in modern therapy. While it is complex to administer, it is also reversable and titratable and not dependent on renal clearance.
The newer direct oral anticoagulants (DOACs) include dabigatran and Xa inhibitors. They both have predictable anticoagulant effects. Dabigatran is associated with GI side effects but is reversible with very significant renal dependence and so a good choice for patients with liver impairment. In contrast Xa inhibitors that are currently available in Canada are irreversible and have less renal dependence. Over time, Xa inhibitors, which have similar pharmacokinetic profiles as LMWH, will be used with increasing frequency, as the availability of a reversal agent may increase comfort. Furthermore, there are data on the efficacy of DOACs in patients with splanchnic vein thrombosis (SVT) showing that in cirrhotic and non-cirrhotic patients there was no significant differences in the rate of bleeding when DOACs were used in comparison with no anticoagulation, LMWH or warfarin in cirrhotic patients.(12)
As time passes and physicians become more comfortable with newly available medications, their use increases and the “eligible population” expands, thus it is suspected that in time DOACs will become the “go to” agents for SVT.
The Rock and the Hard Place of Anticoagulation
Thrombosis requires anticoagulation, however IBD is associated with bleeding, which anticoagulation could exacerbate. Given this clinical dilemma, planning for anticoagulation in patients with IBD should consider some important clinical questions (Figure 1).
- What type of TE does the patient have, and what are the risks of both direct effects of that TE and of acute extension/recurrence?
- What was the impact of potential bleeding?
- Are there modifiable risk factors for bleeding?
- Remember that IV UFH is a perfectly acceptable anticoagulant and is the only completely reversible anticoagulant we currently have

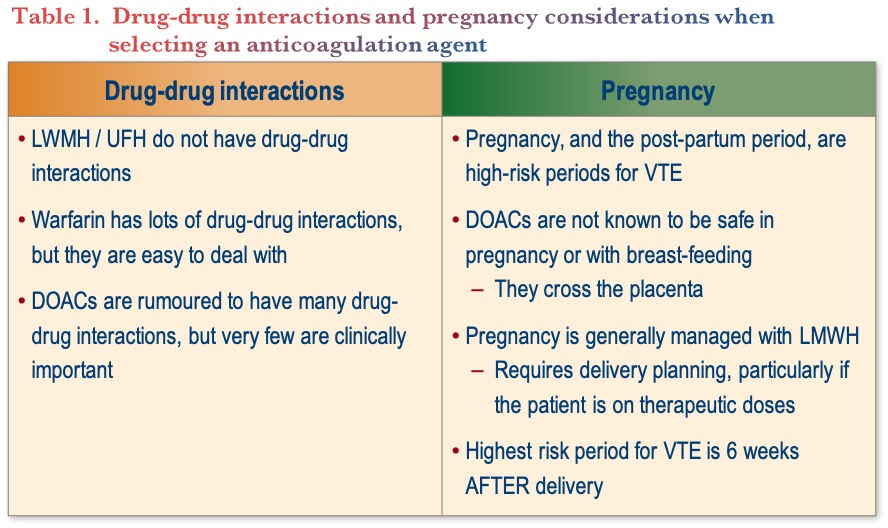
For example, IBD with clinically important VTE is one circumstance for which worsening bleeding may be tolerated as a “cost” of anticoagulation. With respect to surgery, patients who may require immediate surgery should be treated with UFH since it is reversible and does not interfere with anesthetic options. Patients who may require surgery in the future can be treated with either LMWH or a DOAC, as their pharmacokinetics are very similar, and bridging is not required, however warfarin should be avoided. Lastly, there are considerations with respect to drug-drug interactions and pregnancy that need to be considered when selecting an anticoagulation strategy (Table 1).
Pregnancy: A clinical case example
As an example, for considerations related to anticoagulation and pregnancy, this case considers a 24-year-old woman with known UC that presents with a disease flare and progressive jaundice. Despite treatment for her IBD, she reports increasing right upper quadrant pain. The initial ultrasound does not reveal any pathology, but hepatic engorgement leads to a search for hepatic vein thrombosis. She is treated with IV UFH, transitioned to LMWH, then switched to warfarin. She makes a complete recovery in hepatic function and achieves long-term remission of her UC. About two years later, she was having difficulty with laboratory monitoring of the warfarin and was switched to the lower, long-term maintenance dose of a DOAC.
Seven years later, she is pregnant. Given her history of VTE, she is instructed to immediately stop the DOAC and switch to weight-adjusted, therapeutic dose, LMWH. There is no need for routine monitoring during pregnancy except for adjustment based on her weight changes. After a planned delivery she discontinued LMWH 24 hours beforehand to allow the preferred type of anesthesia/analgesia followed by immediate reinstitution of LMWH. She planned on breastfeeding so continues treatment for approximately four weeks. She becomes tired of injecting and switches to warfarin, then plans to return to a DOAC once she finishes breastfeeding.
Conclusions
To summarize, IBD is associated with an increased risk of VTE and ATE due to a procoagulant state that is likely multifactorial. Risk factors may be identified that can be mitigated, while bleeding may need to be tolerated in patients with significant thrombotic burden. For anticoagulation, UFH is preferred in conjunction with control of underlying disease to reduce the risk of thrombosis.
Clinical Case
Initial presentation
- Jolie—46-year-old female who presents to your emergency room with:
- Abdominal cramping
- Worsening bloody diarrhea (10 BMs/day)
- Left leg pain
- Jolie just returned from a 6-week vacation in Indonesia during which she had progressive increase in symptoms
- History of ulcerative colitis
- Had packed her oral 5-ASA therapy in checked luggage, which was subsequently lost
- Only had one-week supply in hand luggage
- She denies sick contacts
- Jolie is on the combined oral contraceptive pill
- Family history of colorectal cancer affecting:
- Her father (who does not have IBD), and
- Her mother who is undergoing treatment for breast cancer
- On examination:
- Afebrile
- BP: 104/56 mmHg
- HR: 102 bpm
- RR: 16 breaths/min
- Oxygen saturation 84% on room air
- Abdomen is tender to palpation, left calf is swollen
- Investigations in ER show:
- WBC: 10.4 x109/L
- Hemoglobin: 109 g/L
- MCV: 82.2 fL
- Platelets: 468 x 109/L
- CRP: 18.2 mg/L
- Creatinine: 104 μmol/L (baseline 70)
- Stools: negative for difficile toxin
- Abdominal X-ray: Diffuse colonic wall thickening, no free air or significant dilation
- D-dimer positive (Note: D-dimer is not a useful test in this setting, as if it was negative the patient should still have further evaluation given her hypoxia and clinical presentation)
- ER doctor consults the GI service
Commentary
- The majority of gastroenterologists would choose to investigate further using a CT pulmonary embolism protocol (CTPE) in this case scenario
Case Evolution
- CTPE confirms a large saddle embolus
- She is too unstable for you to perform a flexible sigmoidoscopy given her high oxygen requirements
Commentary
- For this case, to treat the patient’s pulmonary embolism, the majority of gastroenterologists would start LMWH
- With respect to the patient being on the birth control pill, for their IBD patients, gastroenterologists prefer a progesterone-only option, and consider it needs to be discussed with patients so that they can make the appropriate decisions with regards to contraceptive methods (Note: There is reasonable quality evidence that estrogen containing OCPs can be used safely in fully anticoagulated patients)
Case Evolution
- You liaise with internal medicine who decide to start LMWH
- Jolie’s rectal bleeding worsens
- A day later, Hb is 89 g/L and not entirely explained by volume resuscitation
- Fortunately, her oxygen requirements stabilize enough for you to perform a flexible sigmoidoscopy
- This reveals Mayo 2-3 colitis to beyond the extent of the examination (proximal extent is the descending colon)
Commentary
- As a next step for her IBD, most gastroenterologists would choose methylprednisolone, because, despite active bleeding, they are comfortable using steroids to improve the underlying disease activity
- It is difficult to sort disease vs. treatment contributions to VTE risk
- Steroids increase risk cause of VTE but is multifactorial including underlying disease, inflammatory process, and rarely in medicine does one thing stand alone as the “smoking gun”
- The recent international consensus on the prevention of A/VTE in patients with IBD included the following statements with regards to IBD treatments and risk of VTE:3
- Steroids are associated with an increased risk of A/VTE in patients with IBD
- However, the authors also noted that corticosteroid use can be regarded as a proxy for disease activity, a known risk factor for arterial, thus a causal relationship cannot be fully established
- Anti-TNF agents can be associated with a decreased risk of ATE & VTE in patients with IBD
Case Evolution
- You prescribe methylprednisolone and arrange a biologic work up
- Jolie’s respiratory status stabilizes
- Quantiferon returns negative
- After 72 hours on methylprednisolone, you start infliximab 5 mg/kg
- Internal medicine completes prothrombotic laboratory work up and CT chest/abdomen/pelvis given patient’s family history of malignancy
- Recommends an outpatient mammogram
- You plan outpatient colonoscopy to determine her response to infliximab
Commentary
- With regard to how long to continue anticoagulation, the majority of gastroenterologists would refer to hematology to follow the patient given they are not sure of the optimal duration of therapy
- There is no clear guidance on the optimal duration of treatment, and it is not clear whether the cause of PE is secondary or whether there are ongoing risk factors and therefore whether the patient should stay on long term anticoagulation
- Hematologists would likely be divided as well as to how long to continue anticoagulation – but all would continue it for at least 3 months
- The recent international consensus on the prevention of A/VTE in patients with IBD recommended that prophylaxis be maintained during the inpatient period, followed by the consideration of extended duration of prophylaxis after discharge in patients with strong risk factors for VTE(3)
- Given the safety and efficacy of low dose DOACs, extended therapy should be considered.
- Ongoing risk-benefit ratio assessment is required, particularly with intermittently active disease
- Further studies would be helpful to identify the optimal duration of VTE prophylaxis in particularly high-risk individuals, as well duration of therapy in patients with proven VTE whose IBD has come under control
Case Evolution
- Jolie has excellent response to infliximab with:
- Two to three non-bloody BMs per day
- No abdominal pain
- CRP level of 5 mg/L
- Discharged from hospital on day 9, as she is no longer oxygen dependent
- You complete colonoscopy at 3 months
- She has achieved mucosal healing
- Jolie tells you that the hematologist had previously switched her anticoagulation from LMWH to oral therapy
- She wants you as her gastroenterologist to switch her from intravenous infliximab to oral small molecule therapy, specifically JAK inhibitor tofacitinib
Commentary
- Most gastroenterologists do not want to “rock the boat” in cases like this, thus would not put this patient on JAK inhibitor therapy (tofacitinib, upadacitinib)
- When considering screening for risk factors before initiating JAK inhibitor therapy, it is recommended to ask about personal and family history, and if multiple risk factors are present, then to be cautious
- The recent international consensus on the prevention of A/VTE in patients with IBD included the following information with respect to newer therapies for IBD such as JAK inhibitors(3)
- No increase in the risk of VTE has been observed in the overall UC population treated with tofacitinib based on the evidence to date
- Tofacitinib is not associated with an increased risk of major adverse cardiovascular events (MACE) in patients with UC
- Further evidence is needed regarding the drug-related risk of thrombosis with newer therapies in the IBD population, in addition to the need for specific risk assessment tools for thrombotic complications in patients with IBD
- If it is determined that a JAK inhibitor is the patient’s best disease modifying agent, and there is concern about risk factors, rather than avoiding the optimal IBD treatment based on a very hard to define CV risks, prophylaxis can be provided (e.g., DOAC)
References
-
Yuhara H, Steinmaus C, Corley D, et al. Meta-analysis: the risk of venous thromboembolism in patients with inflammatory bowel disease. Aliment Pharmacol 2013;37:953–62.
-
Zezos P, Kouklakis G, Saibil F. Inflammatory bowel disease and thromboembolism. World J Gastroenterol. 2014;20:13863–
-
Olivera PA, Zuily S, Kotze PG, et al. International consensus on the prevention of venous and arterial thrombotic events in patients with inflammatory bowel disease. Gastroenterol Hepatol. 2021;18:857-73.
-
Landman C, Nahon S, Cosnes J, et al.; Groupe d’Etude Thérapeutique des Affections Inflammatoires du Tube Digestif. Portomesenteric vein thrombosis in patients with inflammatory bowel disease. Inflamm Bowel Dis. 2013;19:582–9.
-
Grainge MJ, West J, Card TR. Venous thromboembolism during active disease and remission in inflammatory bowel disease: a cohort study. 2010;375:657–63.
-
Aarestrup J, Jess T, Cobylecki CJ, et al. Cardiovascular risk profile among patients with inflammatory bowel disease: a population-based study of more than 100 000 individuals. J Crohns Colitis. 2019;13:319–23.
-
Carty M, MacEy M, Rampton DS. Inhibition of platelet activation by 5-aminosalicylic acid in inflammatory bowel disease. Aliment Pharmacol Ther. 2000;14:1169–79.
-
Sehgal P, Colombel J-F, Aboubakr A, et al. Systematic review: safety of mesalazine in ulcerative colitis. Aliment Pharmacol Ther. 2018;47:1597–1609.
-
Higgins PDR, Skup M, Mulani PM, et al. Increased risk of venous thromboembolic events with corticosteroid vs biologic therapy for inflammatory bowel disease. Clin Gastroenterol Hepatol. 2015;13:316–21.
-
Sarlos P, Szemes K, Hegyi P, et al. Steroid but not biological therapy elevates the risk of venous thromboembolic events in inflammatory bowel disease: a meta-analysis. J Crohns Colitis. 2018;12:489–98.
-
Witt DM, Nieuwlaat R, Clark NP, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: optimal management of anticoagulation therapy. Blood Adv. 2018; 2(22):3257–91.
-
Ageno W, Westendorf JB, Contino L, et al. Rivaroxaban for the treatment of noncirrhotic splanchnic vein thrombosis: an interventional prospective cohort study. Blood Adv. 2022;6(12):3569–78.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC AGAF, Director, Division of Gastroenterology, Professor, Department of Medicine, McMaster University, Hamilton, ON
Contributing Author
Mark Crowther, MD MSc FRCPC CAGF AGAF, Chair, Department of Medicine, Leo Pharma Chair in Thromboembolism, McMaster University, Hamilton, ON
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
Eytan Wine, MD PhD FRCPC, University of Alberta, Edmonton, AB
IBD Dialogue 2023·Volume 19 is made possible by unrestricted educational grants from…
![]()
![]()
![]()




![]()

Published by Catrile & Associates Ltd., 167 Floyd Avenue, East York, ON M4J 2H9
(c) Catrile & Associates Ltd., 2023. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.