Unlocking Ultrasound’s Role in IBD: Outcome Prediction, Endpoint Assessment, and Treatment Strategy
Unlocking Ultrasound’s Role in IBD: Outcome Prediction, Endpoint Assessment, and Treatment Strategy
March 31, 2026
Issue 03
Mentoring in IBD is an innovative and successful educational program for Canadian gastroenterologists that includes an annual national meeting, regional satellites in both official languages, www.mentoringinibd.com, an educational newsletter series, and regular electronic communications answering key clinical questions with new research. This issue is based on the presentation made by Dr. Cathy Lu, at the 26th annual national meeting, Mentoring in IBD XXVI: The Master Class, held in Toronto on November 14, 2025.
Introduction
The objectives of this presentation were to review emerging evidence for intestinal ultrasound (IUS) in predicting outcomes in inflammatory bowel disease (IBD), to discuss endpoints when using IUS to treat to target, to identify when IUS can be most beneficial, and to explore how IUS can be used in clinical practice.
There are four key sonographic features of active IBD to consider with IUS.1 The first feature is bowel wall thickness, where an abnormal thickness is defined as >3mm for both Crohn’s disease (CD) and ulcerative colitis (UC). The second feature is the location and length of areas of colour Doppler blood flow, which measures inflammation or hyperaemia. Increased inflammation leads to neovascularization resulting in greater blood flow detection on the Doppler signal and is often accompanied by the “comb sign.” The third feature is the amount of inflammatory fat around the bowel. The last feature is the loss of stratification due to inflammation where the layers of the bowel are more difficult to distinguish.1 These four features have been validated2 and included in the International Bowel Ultrasound-Segmental Activity Score (IBUS-SAS),3 which is frequently used in research and clinical settings for CD and UC. The Milan Ultrasound criteria (MUC) is another assessment criteria frequently used for UC.4
Predictive Value of IUS in Early CD
To assess the predictive value of IUS in early CD, a prospective cohort study followed 201 newly diagnosed patients with CD on stable therapy. IUS and endoscopy were performed every 3 months for one year. Bowel wall thickness and classic inflammatory ultrasound features improved by 3 months. In the ileum, improvements continued throughout the year, whereas those in the colon plateaued after 3 months. Overall, the study showed that 38% of patients achieved transmural remission at 3 months and 41% of patients at 12 months. In addition, achievement of transmural remission at 3 months was associated with achieving corticosteroid-free remission and a lower risk of treatment escalation at all follow-ups within the first year.5
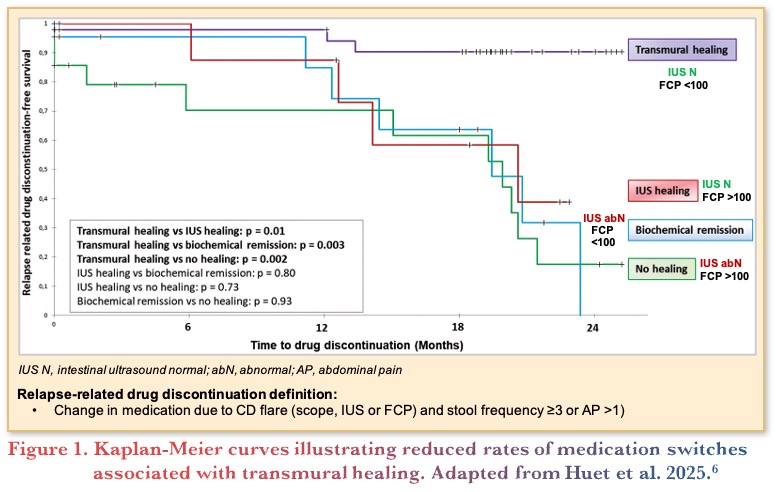
Another prospective study followed 112 patients with CD on therapy where fecal calprotectin (FCP) with concomitant IUS was used to assess mucosal and transmural healing. Patients who achieved transmural healing measured as normalizataion of bowel and color Doppler signal (45%) had a lower risk of requiring medication switches due to flares as shown in Figure 1. Those who only had mucosal remission defined as FCP <100 ug/g but did not achieve transmural healing on IUS, had higher relapse rates than those with both FCP <100 ug/g and transmural healing. Further, transmural healing was a predictor of decreased bowel damage progression (i.e., requirement for surgery or development of strictures, fistulas, or worsening of pre-stenotic dilation).6
In addition, a multicenter retrospective real-world study showed patients with CD who initiated biologic therapy <12 months after diagnosis who achieved transmural remission had decreased bowel damage progression, decreased need for CD-related surgery, and decreased therapy escalation compared to those with late biological therapy initiation (>12 months).7
Taken together, the findings from these studies suggest that transmural remission progressively improves in the terminal ileum over one year. For patients with CD, transmural remission is associated with less steroid use, treatment escalation, drug switches, surgery, and bowel damage, supporting its predictive value in CD.
Predictive Value of IUS in UC
Studies have also investigated the predictive value of IUS in UC. UC is no longer viewed as solely a mucosal disease due to more recent evidence supporting involvement into the deeper layers of the intestinal wall. A pivotal study evaluated IUS as a surrogate marker for endoscopic activity. Thirty patients with moderate-to-severe disease (i.e., Mayo score of 2 or 3) starting tofacitinib underwent a baseline IUS and flexible sigmoidoscopy. The study found that a bowel wall thickness of 2.8 mm at week 8 was strongly associated with a Mayo score of 0 and a 32% reduction in bowel wall thickness, corresponded with an improvement of >1 point on the Mayo score.8
A recent study from 2025 followed 61 patients with UC that were in remission on stable therapy with a Mayo endoscopic score of >1, and had an ultrasound within 6 months. Over the 20-month median follow-up, 72% of patients were found to have transmural healing and a markedly lower risk of relapse compared to those who only achieved endoscopic healing (7.5% versus 29%). Therefore, the investigators concluded that IUS can be used to effectively stratify UC patients by their risk of relapse.9
In another prospective study of 91 patients with UC on stable therapy, the Milan Ultrasound Criteria (MUC) were used as a predictor of transmural healing. Findings showed subjects with a MUC score >6.2 (indicating active disease), had an increased need for drug escalation, steroids, hospitalization, and colectomy.10
Overall, improvements in bowel wall thickness in UC closely reflect the change in the endoscopic Mayo score, reinforcing the value of IUS in monitoring disease activity. A MUC score <6.2, used to reflect transmural healing, is associated with a lower need for drug escalation, steroids, hospitalization, and risk of colectomy. Transmural remission is also associated with a reduced risk of future UC flares, highlighting its importance as a therapeutic target and predictive value in UC.
Role of IUS in Treating-To-Target in IBD
According to an expert consensus statement, transmural remission for both UC and CD is defined as a bowel wall thickness <3mm, normal colour Doppler signal, and a bowel wall thickness up to 4mm for diverticular disease. Treatment response is defined as a decrease in bowel wall thickness by 25%, >2mm, or >1mm and one colour Doppler signal.11 Transmural healing has not yet been evaluated as an outcome in randomized controlled trials (RCTs) in IBD, however a currently ongoing RCT, VECTORS,12 is assessing vedolizumab dose escalation according to treat-to-target parameters with FCP and C-reactive protein (CRP) versus IUS endpoints.
In a sub-study of the STARDUST trial, the effect of ustekinumab on transmural bowel inflammation was assessed over time using IUS. Findings showed that transmural remission improved progressively and could be observed as early as 4 weeks after treatment initiation. Transmural healing was only observed in the terminal ileum for about 13% of patients and 50% of patients in the colon.13
Achieving transmural remission is a favourable outcome but can be challenging to achieve and should not drive repeated cycling through multiple therapies. For some patients, particularly those with strictures, this endpoint may never be attained.
When to Use IUS
IUS is a valuable tool that can be used in nearly all patients with IBD, especially pregnant and pediatric patients. IUS can also help differentiate IBD from irritable bowel syndrome, detect other pathologies, assess early post-operative reoccurrence, and distinguish hot versus cold strictures. Given the long wait times for computed tomography (CT) and magnetic resonance imaging (MRI), and given that patients do not always submit FCP samples, IUS offers a convenient alternative for cross-sectional imaging, providing key information to make timely and appropriate clinical decisions.
As an example of its benefits in pregnant IBD patients, in a multicenter international prospective cohort study, IUS was used to predict adverse obstetric outcomes. IUS was conducted pre-conception, during each trimester, and postpartum. Findings showed that in the second trimester, a bowel wall thickness >6mm with active Doppler signal were associated with a 4-fold increased risk of preterm labour, a 3-fold increased risk of preeclampsia, a 2-fold increased risk of low birth weight at delivery, and a 2-fold increased risk of a neonatal intensive care unit admission.14
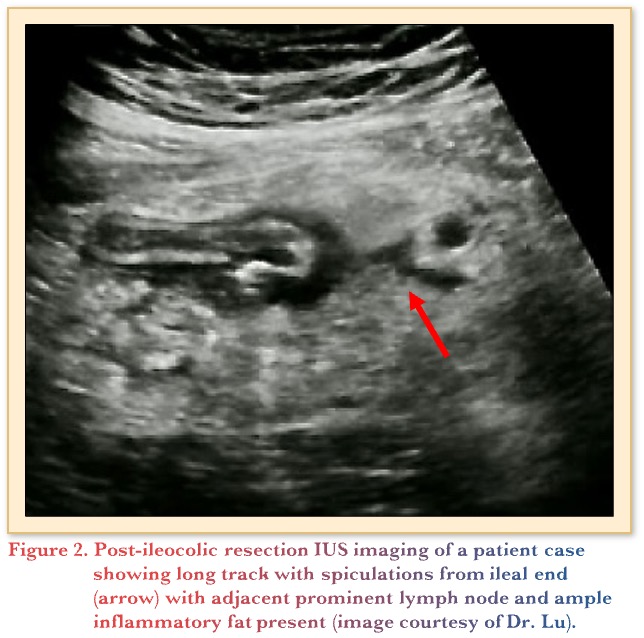
To assess its utility post-operatively, a prospective, multicenter observational cohort study evaluated the use of IUS to detect post-operative recurrence following ileocolic resection. At the 1-year mark, IUS findings were comparable with endoscopy, where wall thickening (>3mm) and FCP >50 µg/g accurately predicted recurrence, with <5% false classifications.15 Below in Figure 2 is an example of an IUS scan of a 45-year-old patient following her second surgery for fibrostenotic CD. She reported abdominal pain 2 weeks post-ileocolic resection with an end-to-side anastomosis. The treating gastroenterologist feared a possible leak. A subsequent CT scan revealed no leak and confirmed an inflammatory mass with a spiculation. The patient continued to improve following antibiotics and was started on risankizumab. Recent follow-up imaging shows complete normalization.
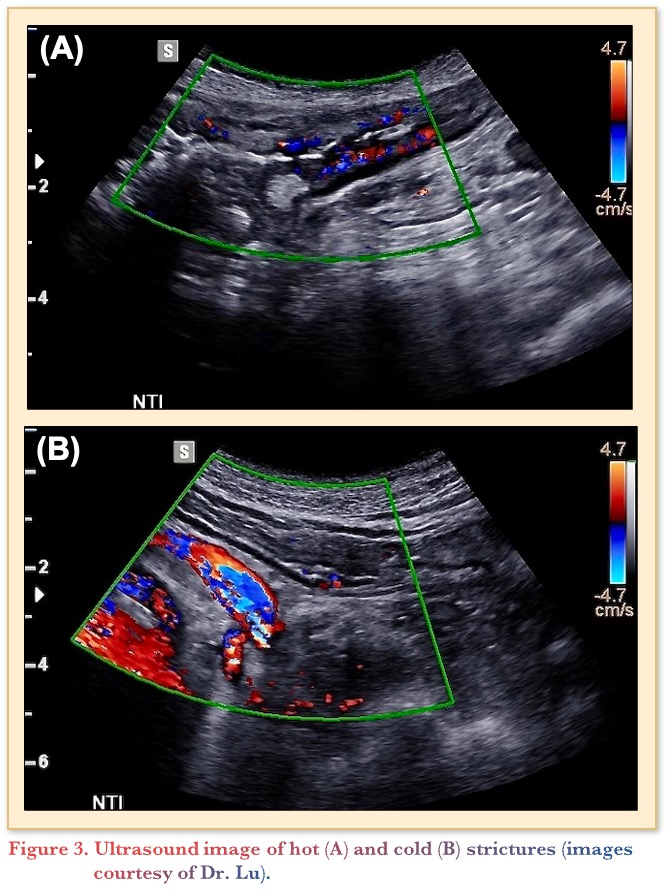
IUS can also be used to discriminate hot and cold strictures. Figure 3 depicts the imaging differences between the two, with a hot stricture (A) and a cold stricture (B). The hot stricture shows ample hyperemia, appearing similar to a string of lights along the bowel wall with prestenotic dilation. The cold stricture is thinner with profound prestenotic dilation covering almost 50% of the screen, with much less signal in the bowel wall itself. This particular patient would benefit from surgery, whereas the patient on the left with the hot stricture could trial additional advanced therapies.
IUS in Clinical Practice
The American College of Gastroenterology (ACG) Clinical Guidelines for UC state that IUS can be used similarly to FCP and CRP to monitor response to therapy.16 The European Crohn’s and Colitis Organization (ECCO) Guidelines recommend the use of IUS or MRE for all newly diagnosed CD patients.17 The ECCO Guidelines also recommend IUS for monitoring at the 12-week mark and prophylactically at 6 to 12 months for patients responding well to treatment. Based on the TRUST&UC study, IUS is recommended at 1–2 weeks, at 6–8 weeks, and as needed to monitor treatment response based on the fact that improvements were observed as early as 2 weeks in the study.18 In clinical practice, responses have been noted as early as 24–48 hours. A recent Delphi by Lu and colleagues presented at ECCO 2026 supports the use of IUS in acute severe UC and recommends that IUS can be used for follow-up at 48 hours following administration of medical therapy.19
Currently, only a limited number of gastroenterologists use IUS in clinical practice. Most formal training is offered through the International Bowel Ultrasound Group; however, the required 4-week hands-on training can be challenging for practicing physicians to schedule. New hybrid training models are being developed. The integration of IUS into gastroenterology and IBD fellowship curricula is being actively explored.
Ultimately, IUS serves as a beneficial tool for predicting clinical outcomes, assessing transmural healing, and offers a noninvasive method of patient evaluation that supports improved quality of care for patients with IBD.
Clinical Case
Emily is a 25-year-old patient with a new diagnosis of Crohn’s disease. She has a known family history of IBD (ulcerative colitis in her father) and presented with abdominal pain, diarrhea (4 non-bloody loose stools per day) and a 5 kg weight loss. She previously had intermittent abdominal pain for several years, which she attributed to stress. She is mildly anemic (Hemoglobin [Hb]=107 g/L and mean corpuscular volume [MCV]=72 fL), with an elevated C-reactive protein (CRP) of 22.1 mg/L and a fecal calprotectin (FCP) of 823 µg/g. Stools were negative for infection (Clostridioides difficile, and Culture and Sensitivity). While waiting to be seen by GI, the family doctor prescribed a short course of prednisone (50 mg x 7 days), which helped improve her symptoms as they were convinced this was IBD. Given the high suspicion for IBD, you performed an urgent colonoscopy (C-scope). C-scope showed ileocolonic disease with deep ulcers extending 5 cm into the ileum and a non-passable stricture located proximally. Aphthous ulcers were also found in the cecum. Remainder of colon was unremarkable. Simple Endoscopic Score for Crohn’s Disease (SES-CD)=14. After her endoscopy, you book an appointment to review results and discuss the diagnosis and treatment plan.
Commentary
- The majority of gastroenterologists do not have access to bedside IUS and are not using it.
- Rather than performing IUS themselves, most physicians will rely on a colleague to perform IUS.
Case Evolution
You decide to ask your colleague trained in IUS to do an IUS to get a baseline assessment
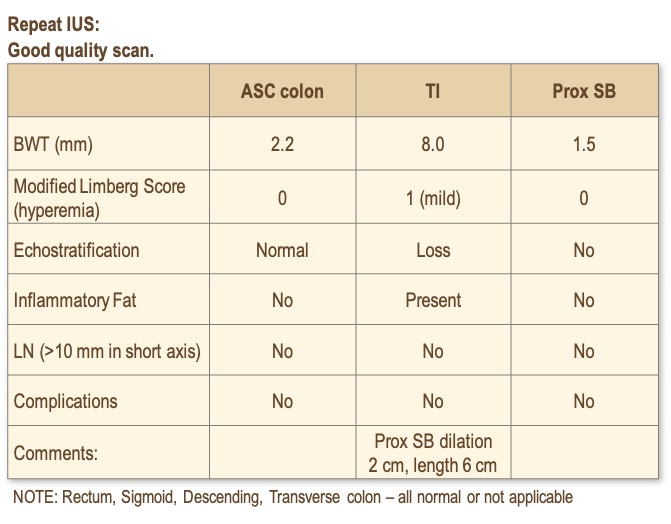
You receive an IUS report that states:
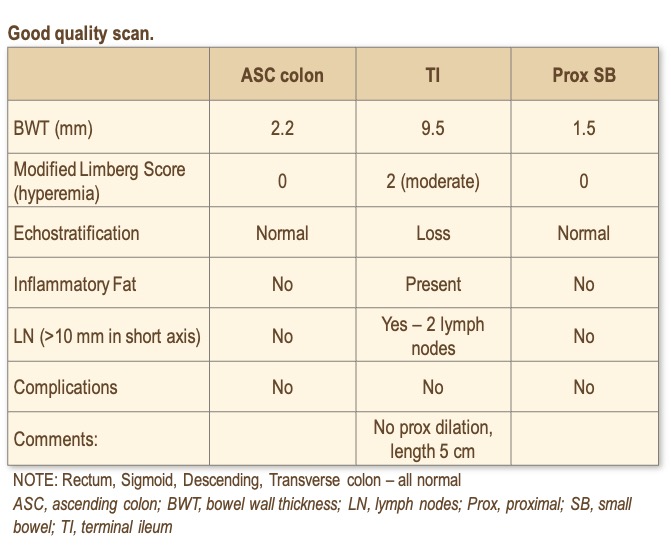
Emily starts on an advanced therapy and is booked for an outpatient follow-up with IUS in 14 weeks. At this time, she requests steroids to help manage her symptoms as the 7-day course did help her. You provide a prescription for a short taper of prednisone (with calcium and vitamin D supplementation).
At the follow-up, she has completed the steroid taper and is starting to have recurrent symptoms of abdominal pain despite the advanced therapy. Her FCP is now 452 µg/g.
Commentary
- Most gastroenterologists would optimize the current treatment dose (or add an immunomodulator) and/or conduct more imaging.
Case Evolution
You dose optimize and request a MRE. There is a several-month wait for MRE for outpatients.
Three months later, the MRE shows a normal colon, but a 6.2 long stricture with mild proximal dilation. There is mild mucosal inflammation but there also appears to be fibrosis. She grumbling symptoms with intermittent abdominal pain (worsened by eating certain foods) and fatigue despite adherence to therapy and a low-fibre diet. She reports some nausea but denies vomiting. Her FCP is now 246 µg/g.
Commentary
- The majority of gastroenterologists would refer to surgery for an ileocecal resection.
- Some suggest switching to advanced therapies, with only a few recommending a repeat IUS.
Case Evolution
You see her with your colorectal surgeon in the IBD Medical Surgical clinic. She admits that she is intermittently smoking cigarettes (<1/2 pack per day). She consents to a minimally invasive ICR. You continue her current therapy as she waits for surgery. She is advised to call if her symptoms worsen. She is counselled on smoking cessation.
She undergoes a laparoscopic ICR. They remove her cecum and 12 cm of ileum. Resection margins are negative for inflammation. Fortunately, she has been able to quit smoking.
Commentary
- There was a divide among the gastroenterologists:
- Many would conduct an IUS. Some gastroenterologists would continue with the current therapy, while others would switch to a different advanced therapy. A smaller group of physicians prefer waiting before making any decisions given she recently quit smoking.
Case Evolution
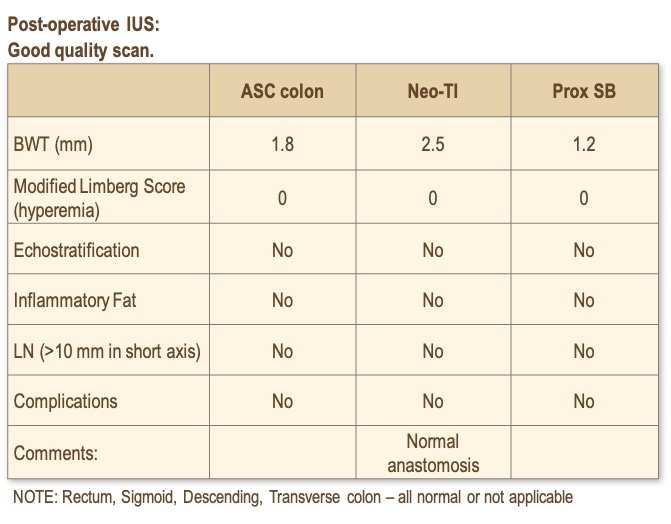
Based upon the Crohn’s and Colitis Canada Clinical Care Pathways, you assess that she has a moderate risk of recurrence and decide to continue her current therapy. At her 6-month follow-up, her FCP is 32 µg/g. She is clinically well, denies abdominal pain and can eat a normal diet. She has not resumed smoking.
Commentary
- Most gastroenterologists would continue therapy and perform a scope.
Case Evolution
Ileocolonoscopy showed Rutgeerts i2a; anastomosis is patent. You continue with her advanced therapy. She continues to feel well. She asks about discontinuing her medications…
References
- Kucharzik T, Kannengiesser K, Petersen F. The use of ultrasound in inflammatory bowel disease. Ann Gastroenterol. 2017;30(2):135–44.
- Lu C, Verstockt B, Winter MW, et al. Review article: extending the frontiers of intestinal ultrasound knowledge, performance and expansion. Aliment Pharmacol Ther. 2026;63(1):40–56.
- Novak KL, Nylund K, Maaser C, et al. Expert consensus on optimal acquisition and development of the international bowel ultrasound segmental activity score [IBUS-SAS]: a reliability and inter-rater variability study on intestinal ultrasonography in Crohn’s disease. J Crohns Colitis. 2021;15(4):609–16.
- Allocca M, Filippi E, Costantino A, et al. Milan ultrasound criteria are accurate in assessing disease activity in ulcerative colitis: external validation. United Eur Gastroenterol J. 2021;9(4):438–42.
- Madsen GR, Wilkens R, Attauabi M, et al. Intestinal ultrasound as a prognostic tool in new-onset ulcerative colitis-a Copenhagen IBD Cohort Study. J Crohns Colitis. 2025;19(4):jjaf033.
- Huet J, Mathieu K, Dodel M, et al. Transmural healing assessed by combination of fecal calprotectin and intestinal ultrasonography is associated with reduced risk of bowel damage progression in patients with Crohn’s disease. Inflamm Bowel Dis. 2025;31(10):2767–76.
- Revés J, Fernandez-Clotet A, Ordás I, et al. Early biological therapy within 12 months of diagnosis leads to higher transmural healing rates in Crohn’s disease. Clin Gastroenterol Hepatol. 2025;23(7):1194–203.e2.
- de Voogd F, van Wassenaer EA, Mookhoek A, et al. Intestinal ultrasound is accurate to determine endoscopic response and remission in patients with moderate to severe ulcerative colitis: a longitudinal prospective cohort study. Gastroenterology. 2022;163(6):1569–81.
- Lim CT, Teichert C, Pruijt M, et al. Transmural healing in ulcerative colitis patients improves long-term outcomes compared to endoscopic healing alone. J Crohns Colitis. 2025;19(9):jjaf149.
- Allocca M, Dell’Avalle C, Craviotto V, et al. Predictive value of Milan ultrasound criteria in ulcerative colitis: a prospective observational cohort study. United Eur Gastroenterol J. 2022;10(2):190–7.
- Ilvemark JFKF, Hansen T, Goodsall TM, et al. Defining transabdominal intestinal ultrasound treatment response and remission in inflammatory bowel disease: systematic review and expert consensus statement. J Crohns Colitis. 2022;16(4):554–80.
- NCT06257706. VECTORS – A study to evaluate transmural healing as a treatment target in Crohn’s disease (VECTORS). Alimentiv Inc.; January 15, 2026. Accessed February 16, 2026. https://clinicaltrials.gov/study/NCT06257706
- Kucharzik T, Wilkens R, D’Agostino MA, et al. Early ultrasound response and progressive transmural remission after treatment with ustekinumab in Crohn’s disease. Clin Gastroenterol Hepatol. 2023;21(1):153–63.e12.
- Prentice RE, Flanagan EK, Wright EK, et al. Active inflammatory bowel disease on intestinal ultrasound during pregnancy is associated with an increased risk of adverse pregnancy and neonatal outcomes independent of clinical and biochemical disease activity. Gastroenterology. 2025;169(4):647–62.
- Furfaro F, D’Amico F, Zilli A, et al. Noninvasive assessment of postoperative disease recurrence in Crohn’s disease: a multicenter, prospective cohort study on behalf of the Italian group for inflammatory bowel disease. Clin Gastroenterol Hepatol. 2023;21(12):3143–51.
- Rubin DT, Ananthakrishnan AN, Siegel CA, Barnes EL, Long MD. ACG Clinical Guideline Update: Ulcerative Colitis in Adults. Am J Gastroenterol. 2025;120(6):1187–224.
- Kucharzik T, Taylor S, Allocca M, et al. ECCO-ESGAR-ESP-IBUS Guideline on Diagnostics and Monitoring of Patients with Inflammatory Bowel Disease: Part 1. J Crohns Colitis. 2025;19(7):jjaf106.
- Maaser C, Petersen F, Helwig U, et al. Intestinal ultrasound for monitoring therapeutic response in patients with ulcerative colitis: results from the TRUST&UC study. Gut. 2020;69(9):1629–36.
- Lu C, et al. Unpublished data.
Editor-in-Chief
John K. Marshall, MD MSc FRCPC CAGF AGAF
Professor, Department of Medicine
Director, Division of Gastroenterology
McMaster University
Hamilton, ON
Contributing Author
Cathy Lu, MD, MSc
Associate Professor, Cumming School of Medicine
Department of Medicine | Gastroenterology
University of Calgary
Calgary, AB
Mentoring in IBD Curriculum Steering Committee
Alain Bitton, MD FRCPC, McGill University, Montreal, QC
Karen I. Kroeker, MD MSc FRCPC, University of Alberta, Edmonton, AB
Cynthia Seow, MBBS (Hons) MSc FRACP, University of Calgary, Calgary, AB
Jennifer Stretton, ACNP MN BScN, St. Joseph’s Healthcare, Hamilton, ON
Eytan Wine, MD PhD FRCPC, University of Toronto, Toronto, ON
IBD Dialogue 2026·Volume 22 is made possible by unrestricted educational grants from…
![]()
![]()









Published by Published by Kalendar Inc.., 7 Haddon Avenue, Scarboro, ON M1N 2K7
(c) Kalendar Inc. 2026. All rights reserved. None of the contents may be reproduced in any form without prior written permission from the publisher. The opinions expressed in this paper are those of the authors and do not necessarily reflect the opinions or recommendations of the sponsors, the grantor, or the publisher.
